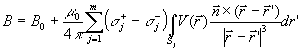|
Multimodal Integration of High Resolution EEG, MEG and
Functional Magnetic Resonance Data
F. Babilonia, F. Carduccia, C. Del Grattab, G.M. Roberti, F. Cincottia,
O. Bagnia,
G.L. Romanib, P.M. Rossinic , C. Babilonia
a2° Cattedra di Biofisica-Istituto di Fisiologia Umana,
Università di Roma ‘La Sapienza’,
Roma, Italy;
E-mail: babilonif@axrma.uniroma1.it
bDipartimento di Scienze Cliniche e Bioimmagini, Istituto di
Tecnologie Avanzate Biomediche Università "G. D’Annunzio", Chieti, Italy;
E-mail: glromani@dns.unich.it
c A.Fa.R-IRCCS "San Giovanni di Dio" Istituto Sacro
Cuore di Gesù, via dei Pilastroni 4,
Brescia, Italy;
cA.Fa.R. CRCCS - Divisione di Neurologia, Osp. FBF Isola Tiberina, Roma, Italy;
E-mail: rossini.pm@mclink.it
Abstract.
Two advanced technologies for the integration of multimodal
electroencephalographic (EEG), magnetoencephalographic (MEG), and functional magnetic
resonance (fMR) data are proposed. These technologies include high surface sampling of
EEG-MEG data, realistic MR-constructed subject’s multi-compartment (scalp, skull,
dura mater, cortex) head model, multi-dipole source model, and regularized linear inverse
source estimate based on boundary element mathematics. Linear inverse source estimates of
cortical electrical activity were regularized not assuming that covariance of background
electromagnetic noise between sensors was zero. Furthermore, fMR data were used as a
constraint for the linear inverse source estimate from highly sampled (128 channels) EEG
data. Linear inverse source estimate from the only EEG data served as a reference. The
proposed technologies modeled cortical activity related to voluntary right finger
movements. Modeling of movement-related cortical activity showed higher spatial
information content from the combined EEG and MEG data than from these data considered
separately. During the preparation and execution of the movement, the combined EEG-MEG and
EEG-fMR linear inverse solutions modeled a bilateral, contralaterally preponderant,
activation of primary sensorimotor cortex as well as the activation of supplementary motor
area. These results support the hypothesis that bilateral human M1-S1 is involved not only
in the preparation but also in the execution of unilateral distal movements. In
conclusion, multimodal EEG-MEG-fMR linear inverse source estimate is a powerful tool to
satisfactory localize and describe the temporal evolution of the human event-related
cortical activity.
1. Introduction
An ideal technology for the study of the human brain activity would
have high spatial-temporal resolution to reveal the rapid evolution of patterns of
event-related cortical activation. At the moment, such a technology does not exist. Thus,
there is increasing interest in the combination of neuroimaging techniques having high
temporal but low spatial resolution (i.e. electroencephalography, EEG and
magnetoencephalography MEG) with those having high spatial but low temporal resolution
(positron emission tomography, PET and functional magnetic resonance, fMR). Combined
EEG and MEG data increase stability and accuracy in the solution of the linear inverse
source estimate of human event-related electromagnetic cortical activity. To date, the
linear inverse source estimate from combined EEG and MEG data has been based on (i)
preliminary normalization by the covariance of background electromagnetic noise; (ii)
realistic MR-constructed head model; (iii) multi-dipole source model; (iv) regularization
procedure; and (v) boundary or finite element mathematics (Fuchs et al., 1998). Commonly
used regularization schemes assume that between-sensor covariance of background
electromagnetic noise is zero, although this assumption rarely holds in the real case. The
rationale for the combined use of EEG and MEG data in the linear inverse source estimate
is that the MEG activity is poorly contaminated by head volume conduction effects and
subcortical fields, while the EEG (but not MEG) is sensitive to the activation of radial
cortical sources (Kristeva et al., 1991). The use of fMR as a constraint in the linear
inverse source estimate of EEG or MEG data is a promising approach to improve the spatial
information content of the source EEG-MEG solutions. In the present study, linear inverse
source estimates from combined EEG and MEG data were computed using realistic head and
source models, boundary element mathematics, and a regularization scheme taking into
account that between-sensor covariance of background electromagnetic noise can be nonzero.
Furthermore, fMR data were used as a constraint in the linear inverse source solution of
the EEG data. These technologies were used to re-evaluate the controversial hypothesis
that bilateral primary sensorimotor areas (M1-S1) are involved in both the preparation and
execution of voluntary unilateral finger movements.
2. Methods
2.1 Subjects and task
The present study was carried out on two healthy, right-handed
(Edinburgh Inventory) male volunteers. The experiments were undertaken with the
understanding and written consent of each participant. General procedures were approved by
the local institutional ethics committee. During the experiments, subjects lay on a
non-magnetic wooden bed placed in a dimly-lit, sound-damped, and magnetically-shielded
room (Vacuumschemelze, Hanau). Head was stabilized by a vacuum cast. Motor task consisted
of brisk, internally triggered unilateral right middle finger extensions followed by
passive return to the original resting position (inter-movement interval: 2-5 sec).
Subjects were asked to avoid blinking, eye movements, and respiration immediately before
and during the movement. A brief training was performed to render stable and reproducible
the motor performance. During these sessions, surface electromyographic (EMG) activity of
extensor digitorum muscle of both sides was monitored (Ag-AgCl cup electrodes; 1-100 Hz
bandpass; 400 Hz sampling rate) to control operating muscle response and involuntary
mirror movements. Furthermore, surface EMG activity of bilateral axial and proximal
muscles was also recorded from the two participants to monitor co-activation of these
muscles in concomitance with the finger movement. No notable co-activation of axial and
proximal muscles was observed.
2.2 EEG recordings
EEG activity was recorded (0.1-100 Hz bandpass) with 128
electrodes (linked earlobe electric reference) during the movement. Electrode positions
and reference landmarks were digitized for subsequent integration between the EEG, MEG,
and MR data. Electrooculogram (0.1-100 Hz passband) and electromyogram (EMG, 1-100 Hz
passband) from m. extensor digitorum of both sides were also recorded. All data
were acquired (400 Hz sampling rate) from 3 sec before to 1 sec after the onset (zerotime)
of the EMG response in the operating muscle. About 200 single trials were collected.
Artifact-free (eye and/or mirror movements) single trials were averaged with respect to
zerotime.
2.3 MEG data acquisition
Movement-related MEG activity was recorded (0.16-250
Hz passband), in separate blocks, from the left and the right hemisphere by a dewar
(diameter: 16 cm) including an array of 25 sensors. The sensor array was centered on C3 or
C4 site of the 10-20 international system, which are roughly overlying the hand
representation of the left and right M1-S1, respectively. This array comprised 9
magnetometers with a 80 mm2 integrated pick-up
coil (plus 3 reference channels to be used for noise cancellation) and 16 axial
gradiometers (250 mm2 area, 8 cm baseline).
The noise spectral density of each sensor channel was 5-7 fT/vHz at 1 Hz. Vertical and
horizontal eye movements were controlled by recording electrooculographic activity
(0.16-250 Hz bandpass) from a pair of Ag-AgCl cup electrodes placed laterally and medially
just above and below the right eye, respectively. The EMG activity of the extensor
digitorum muscle of both sides was recorded as in the training session (see above). Right
finger movements were performed in blocks lasting about 10 min (10 min inter-block
interval). All data were gathered in continuous mode (1000 Hz sampling rate). Positions of
the sensor array with respect to subjects anatomical landmarks (nasion, inion, and
preauricular points) were detected after 1 or 2 recording blocks, in order to register
subtle head movements across the experimental session. Positions of these fiducial
landmarks were also digitized off-line for the subsequent integration between MEG and MR
data.
2.4 Realistic head and source models
Sixty-four T1-weighted sagittal MR images were acquired (30 msec
repetition time, 5 msec echo time, and 3 mm slice thickness without gap). These images
were processed with contouring and triangulation algorithms for the construction of
subject’s head model. The MR-constructed head model reproduced scalp, skull, and dura
mater surfaces with 910 triangles for each surface. Source model was built with the
following procedure: (i) the points belonging to the MR images of the cortex were selected
with a semiautomatic procedure (thresholding algorithm); (ii) these points were subsampled
from 12,000-14,000 to 2,400-4,100; however, the general features of the neocortical
envelope were well preserved especially in correspondence of pre- and postcentral gyri and
frontal mesial area (mesh refinement), (iii) the subsampled points were triangulated
(5,000-7,000 triangles); and (iv) an orthogonal unitary equivalent current dipole was
placed in the center of each triangle forming the cortex compartment. Regions of interest
were drawn on the reconstructed cortical surface to model supplementary motor area (SMA)
and left and right M1-S1.
2.5 FMRI acquisition and analysis
FMR images were acquired from the two volunteers by
means of a Siemens Magnetom Vision 1.5 T scanner equipped with 25 mT/m gradients. The
motor task consisted of repetitive right middle finger extensions at variable frequency
rate (0.5-2 Hz) to make similar the act to that performed during the EEG and MEG
recordings. The experimental design included the acquisition of 64 volumes of fMR for the
rest-movement-rest cycle. The temporal resolution of each volume acquisition was of 5 sec.
Influences of signal intensity drift and head motion on fMR were corrected by
normalization and automated image registration algorithms, respectively. The fMR images
were co-registered with anatomic images of the subjects cortex, which were obtained by the
acquisition of T1-weighted conventional spin-echo-axial-oblique sequence. Percent change
of fMR signal intensity due to the movement was computed for each voxel according to the
procedure of Kim and co-workers (PC map; Kim et al., 1993 a,b). The difference between the
mean rest and movement-related signal intensity was calculated voxel-by-voxel. The
rest-related fMR signal intensity was obtained by averaging the pre-movement and recovery
fMR. Bonferroni-corrected Students t-test was used to minimize alpha inflation effects due
to multiple statistical voxel-by-voxel comparisons (Type I error; p<0.05). Only voxels
with a statistically significant PC activation were considered. The fMR values of these
voxels were assigned and summed by a minimum distance projection algorithm to the nearest
dipoles placed onto the modeled cortical surface.
2.6 Electrical forward solution
Let a head model be constituted by electrically
homogeneous and isotropic compartments simulating scalp, skull, and dura mater. The
forward solution specifying the potential distribution (V) on these compartments Sk (k = 1,...3) due to a dipole is given by the Fredholm
integral equation of the second kind
| |
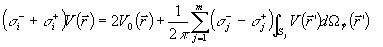 |
(1) |
with
| |
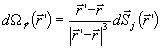 |
(2) |
where
(i) 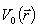 is the potential due to a dipole located in an infinite homogeneous medium;
(ii) is the potential due to a dipole located in an infinite homogeneous medium;
(ii) 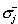 is the conductivity inside the surface S of the multicompartment head model;
(iii) is the conductivity inside the surface S of the multicompartment head model;
(iii) 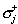 is the conductivity outside the surface Sj;
(iv) m is the total number of compartments within the head model; and
(v) is the conductivity outside the surface Sj;
(iv) m is the total number of compartments within the head model; and
(v) 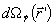 is the solid angle subtended by the surface element dS located in is the solid angle subtended by the surface element dS located in
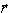 (point of observation (point of observation
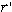 ).
A numerical solution of the Fredholm integral equation can be obtained by decomposing the
surfaces Sk (k = 1,...3) into triangle panels and by using boundary element techniques
(Meijs et al 1993). With the boundary-element techniques, a discrete version of the
Fredholm integral equation is given by ).
A numerical solution of the Fredholm integral equation can be obtained by decomposing the
surfaces Sk (k = 1,...3) into triangle panels and by using boundary element techniques
(Meijs et al 1993). With the boundary-element techniques, a discrete version of the
Fredholm integral equation is given by
where the elements of matrix Ω , vector v,
and vector g are defined as follows: (i) vi is
the potential value in the center of mass of the i-th triangle; (ii) gi is the potential value generated by a source in the
center of mass of the i-th triangle; and (iii) Ωij is the matrix element proportional to the solid angle
subtended by the j-th triangle at the center of mass of the i-th triangle. The numerical
solution of the Fredholm integral equation can be improved using the correction proposed
by Hämäläinen and Sarvas (1989). The linear system of equation 3 is singular since the
potential distribution generated on the scalp compartment by an equivalent dipole
is determined up to a constant. This singularity can be removed by using a
deflation procedure that yields the potential distribution (V) on the compartment surfaces
Sk (k = 1,...3).
2.7 Magnetic forward solution
The magnetic field of a current dipole in a piecewise
homogenous conducting medium is given by
| |
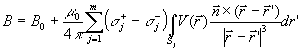 |
(4) |
where (i) B0 is the field of the current dipole in the free
space with each surface integral extending to an interface between homogeneously
conducting media; (ii) n is a unit vector orthogonal to the surface Sj; and
(iii) the summation index j runs over the surfaces. The surface integrals take into
account the effects of volume currents. Notice that these currents are equivalent to
layers of current dipoles orthogonal to the interfaces, with dipole moment per unit area
V(σ+- σ-). The magnetic lead field
matrix was calculated by adding, for each dipole, the free space term and the
corresponding volume current term. Since a boundary element model was used, the volume
currents were modeled by an array of fictitious dipoles, located at the centroids of each
triangle of the reconstructed surfaces, with moment V(σ+- σ-)
times the triangle area
2.8 Integrated electric and magnetic solution
The forward solution specifying the potential scalp field due to an
arbitrary dipole source configuration was computed on the basis of the linear system
where A is the matrix composed of the electric and magnetic lead
fields, b is the measurement array of EEG and MEG data and x is the array of the
unknown cortical dipole strengths.
2.9 EEG-MEG regularization
Since the number of dipoles was much higher than the spatial
measurements, the linear system of equation 5 has infinite solutions. However, a reliable
solution of this system can be obtained with a priori assumption of a minimum energy
constraint. The general formulation of the linear inverse problem based on this assumption
is
| |
X = argmin (||D(Ax-b)|| 2+ λ2 ||C x||2) |
(6) |
where D is the sensor equalization matrix obtained by the Cholesky
decomposition of the inverted covariance matrix S of the EEG and MEG sensors (DTD = S-1)
and C is the matrix that regulates how each EEG or MEG sensor is influenced by dipoles
located at different depths into the source model. The covariance matrix S was derived
from the EEG and MEG data by maximum likelihood estimation on a set of EEG and MEG data
with maximum background electromagnetic noise (i.e., no event-related electromagnetic
signal). The matrix C was given by:
where Wii is a diagonal matrix in which the i-th element is
equal to the norm of the i-th column of the normalized lead field matrix DA, and V is the
matrix of the principal dipole components obtained by the singular value decomposition of
the normalized lead field matrix DA (Fuchs et al 1998). An optimal regularization
of the linear system Ax = b was obtained by the L-curve approach (Hansen 1992). The
L-curve plots the residual norm versus the solution norm at different lambda (λ) values.
Computation of the L-curves and optimal λ correction values were performed by original Hansen’s
routines. With equation 7, an optimal regularization value of 1 was always found (Fuchs et
al 1998).
2.10 EEG-fMR regularization
For the source space metric computation, we normalized
the electric lead field matrix by the column norm, to balance the much more visibility of
the superficial than deep cortical sources of the EEG data. Furthermore, the column
normalization for each modeled cortical source accounted for the movement-related
percentage intensity values (α)
of the integrated fMR data (Kim et al., 1993 a,b). The metric for the source space was
associated with a diagonal matrix C, whose the i-th term was
| |
Cii = ||Ai||2 * g (αi)-2 |
(8) |
where ||Ai|| is the norm of the i-th column of the lead field matrix A
and g(αi) is a function of the
statistically significant percentage increase of the fMR signal, assigned to the i-th
dipole of the modeled source space. The g(αi) function was expressed by
| |
g(αi) = 1 + K αi; αi >0; |
(9) |
where the factor K tuned fMR solutions in the source space for the time
varying electromagnetic component b. With eq. 9, high K value (i.e., Kαi about 10) produced terms Cii, which were
roughly one order of magnitude lower than those used by taking into account the only
column normalization. On the other hand, a low K value (i.e., Kαi <<1) resulted in a Cii value roughly proportional
to the squared column norm, which completely disregarded the fMR solution (i.e., 0% fMR
solution or 100% EEG solution). Optimal value of the K factor was obtained by using the
indexes proposed to evaluate the quality of cortical re-constructions in the linear
inverse problem (Grave de Peralta et al., 1997).
3. Results
Fig. 1 plots mean MEG and EEG wave forms computed in a
subject from the lateral-frontal and medial-parietal areas of both hemispheres in
association with the right finger movement. During the movement preparation, the MEG wave
forms were represented by a bilateral, contralaterally preponderant slow magnetic shift,
starting at about -0.5 sec and culminating at about the zerotime (readiness field-motor
field peak; RF-MFp). This complex showed contralateral lateral-frontal positivity (outward
current flow)/medial-parietal negativity (inward current flow) and ipsilateral
lateral-frontal negativity/medial-parietal positivity. The MEG wave forms presented also a
transient magnetic shifts peaking at about +110 msec (motor evoked field 1 peak, MEF1p).
With respect to the RF-MFp, the MEF1p was higher in amplitude and had reversed polarity
over the contralateral hemisphere. Furthermore, it had low amplitude and non-reversed
polarity over the ipsilateral hemisphere (i.e. negativity). On the other hand, the EEG
wave forms were characterized by a slow negative shift starting at about -1 sec and
peaking close to the movement onset in the frontal, central, and parietal leads (readiness
potential peak, RPp). The analogous of the MEF1p was the component peaking at about +110
msec (movement-related response 1 peak, MRR1p).
The mean amplitude maps of Fig. 2 show the topography of the subject's
cortically projected electromagnetic activity illustrated in Fig. 1 (recorded data). An
intense, dipolar contralateral lateral-frontal and medial-parietal MEG field was observed
at RF-MFp and MEF1p, the MEF1p being reversed in polarity compared to the RF-MFp. In
addition, RF-MFp presented a low-amplitude MEG field reversed in polarity over the
ipsilateral lateral-frontal and medial-parietal areas. The corresponding raw EEG potential
distributions were characterized by large and distant negative and positive maxima,
preponderant in the side contralateral to the movement. The electric field was tilted of
90° with respect to the magnetic field. Amplitude 3-D maps of Fig. 3 illustrate linear
inverse source estimates from EEG, MEG, and combined EEG-MEG data shown in previous
figures. These estimates for the movement preparation and execution periods mapped the
strength of the dipoles used as a cortical source model. There were circumscribed zone of
negativity and positivity in cortical regions roughly corresponding to the M1-S1 of both
sides and SMA. The linear inverse source estimate of the combined EEG-MEG data could
integrate in a unique solution features of linear inverse source estimate of these data
considered separately. Similar results were observed in the other subject.
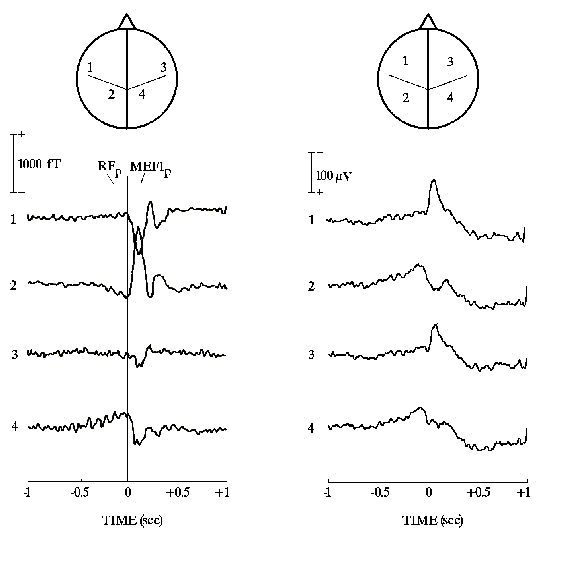
Figure 1. Wave forms of MEG (left) and EEG (right) activity peaking
(p) in the contralateral and ipsilateral hemispheres of a subject during the pre-movement
(readiness field-motor field peak, RF-MFp; readiness potential peak RPp) and performance
(movement-evoked field 1 peak, MEF1p; movement-related response 1 peak, MRR1p) periods of
voluntary unilateral right middle finger extension.
Fig. 4 shows the time evolution (wave forms) of the cortical activation
as indicated by linear inverse source estimates in the subject's regions of interest (M1,
S1, SMA). FMR data were used as a constraint in the linear inverse source estimation. A
map of the linear inverse source estimate at MRR1p (+110 msec) is also shown. This map
represents the linear inverse source estimate forwarded over the subject's dura mater
model. Modeled wave forms of the cortical activity peaked at +115 msec in the S1
contralateral to the movement. Such an activity peaked few msec later in the SMA and
ipsilateral M1 and S1.
Figure 2: Movement-related EEG and MEG activity recorded (128 and
50 channels respectively) about 110 msec after the onset of electromyographic response
accompanying the movement. Color maps of the recorded electric potentials and magnetic
fields are projected over the subject's MR-constructed cortical model for illustrative
purposes. Percent color scale (256 values) is normalized with reference to the maximum
amplitude calculated for each map. Maximum negativity (-100%) is coded in violet and
maximum positivity (+100%) is coded in red.
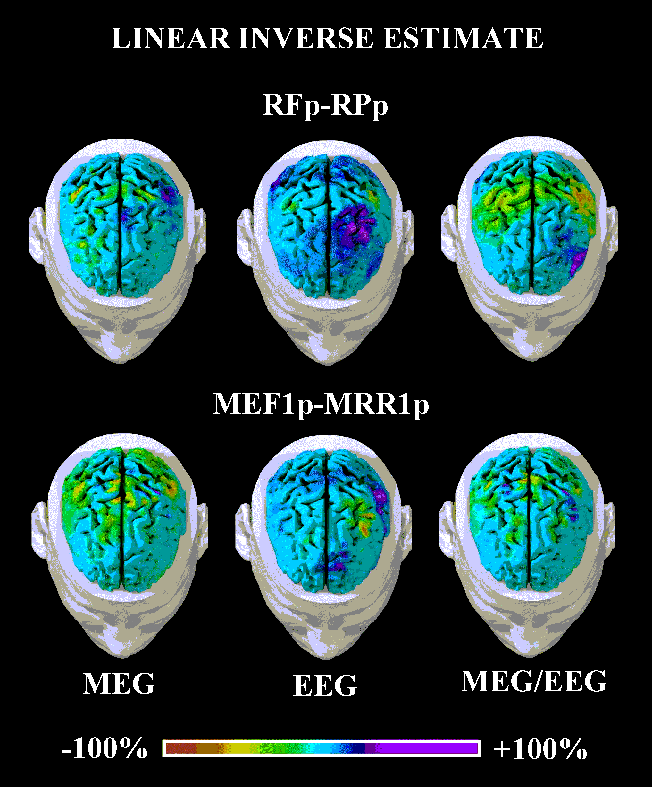
Figure 3: Linear inverse source estimates from EEG, MEG, and
combined EEG-MEG data shown in figure 2. These estimates for the movement preparation
(RF-MFp, RPp) and execution (MEF1p, MRR1p) periods mapped the strength of the dipoles used
as a cortical source model. Percent color scale (256 values) in which maximum negativity
(-100%) is coded in red and maximum positivity (+100%) is coded in violet.
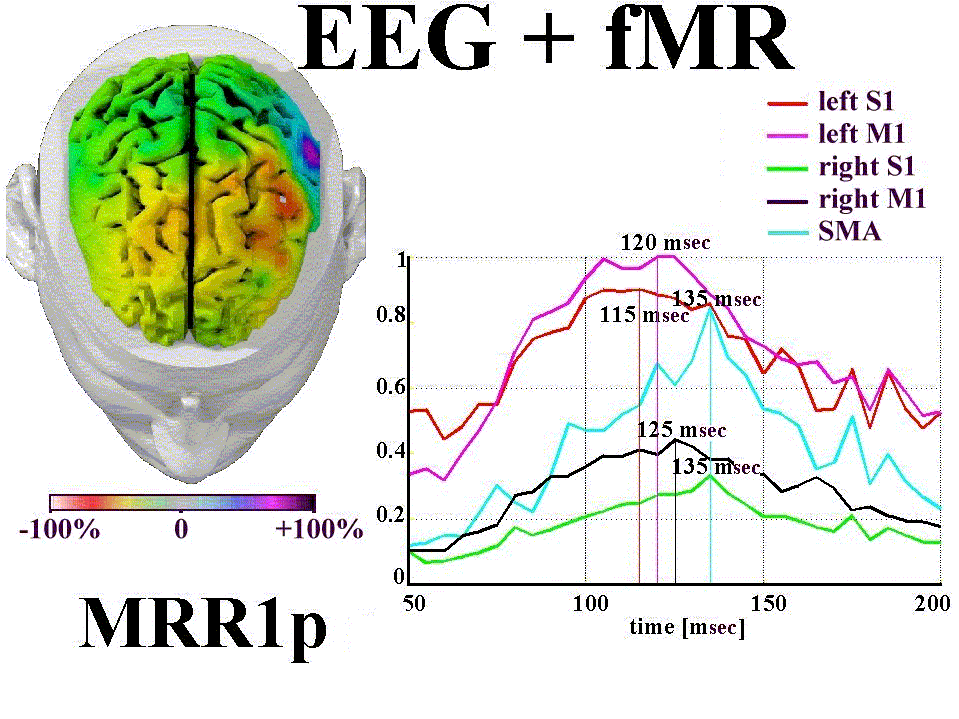
Figure 4. Wave forms of the linear inverse source
estimates of MRR1p computed in the subject's cortical regions of interest (M1, S1, SMA) by
using fMR data as a constraint. The illustrated color map represents this estimate
forwarded over the subject's MR-constructed dura mater model. Percent color scale
(256 values) is used.
4. Discussion
This study presented performances of advanced neuroimaging techniques
for the modeling of human movement-related cortical activity from combined EEG, MEG, and
fMR data. Linear inverse source estimates of the combined EEG and MEG data were
regularized not assuming that between-sensors noise covariance was zero. The
regularization scheme extended to the linear inverse source problem for the underestimated
cases (more dipoles than sensors) the mathematical approach recently used for the
overestimated case (more sensors than dipoles) (Fuchs et al., 1998). Linear inverse source
estimates of the combined EEG and MEG data enhanced spatial information content when
compared to those obtained from the EEG and MEG data considered separately. In fact,
combined EEG and MEG linear inverse source estimates represented in a unique solution
spatial features presented in the EEG and MEG linear inverse source estimates considered
separately. The results of the present study modeled a maximum activation in the
contralateral M1-S1 and in the mesial-frontal cortical area (including SMA). In addition,
a minor activation was modeled in the ipsilateral M1-S1 supporting the working hypothesis
of a bilateral M1 S1 activation during the preparation and execution of unilateral finger
movements. Plausibly, the activation of the ipsilateral M1-S1 during the movement
execution is mainly related to the processing of movement-evoked somatosensory information
supplied by double crossed and uncrossed pathways (Urbano et al., 1996). Putative double
crossed pathway would include dorsal-column lemniscal system and transcallosal M1-S1
connections (Rouiller al., 1994; Wiesendanger et al., 1996). Whereas, putative uncrossed
pathways to ipsilateral M1-S1 may comprise spinoreticular, spinomesencephalic, and
spinocerebellar pathways (Scheibel, 1984). The ipsilateral M1-S1 activation accompanying
the movement execution would subserve transcallosal inhibition of the small uncrossed
pyramidal pathway originating from the contralateral M1-S1 (Sadato et al., 1997). This
M1-S1 activation might also provide some additional control of the operating hand by means
of ipsilateral uncrossed pyramidal pathway (Kristeva et al., 1991). The view of an
ipsilateral finger motor representation in M1-S1 is in line with transcallosal M1-S1
connections, uncrossed (about 10%) pyramidal fibers (Wiesendanger et al, 1996), single
neuron recordings in monkeys (Tanji et al., 1988), transcranial magnetic stimulation in
normal subjects and patients with callosal dysfunction (Meyer et al., 1995; Wassermann et
al., 1994). Furthermore, this view is in line with clinical (Haaland et al, 1981, 1989;
Jones et al, 1989) and PET (Chollet et al., 1991; Weiller et al., 1996) studies in
unilateral hemisphere stroke patients. In contrast, some PET (Colebacht et al., 1991;
Roland et al., 1980; Shibasaki et al., 1993) and functional MR (Boecker et al., 1994; Rao
et al., 1993, 1995) scans in normal subjects showed no involvement of the ipsilateral
M1-S1 in the motor control. A possible explanation for this is that these neuroimaging
techniques may have an insufficient sensitivity for a stable detection of slight M1-S1
signal as that related to stereotyped ipsilateral movements. In fact, PET and fMR scans
showed ipsilateral M1 and S1 activation only when the motor task consisted of controlled
thumb-ulnar finger opposition sequences (Boecker et al., 1994; Kim et al., 1993 a,b;
Schroeder et al., 1995).
In conclusion, the proposed neuroimaging technologies used highly
sampled EEG and MEG data, realistic MR-constructed subject's head models, fine cortical
source model, and fMR data as a physiological constraint of the linear inverse source
estimate. These technologies provided evidence in favor of the hypothesis that in
right-handed subjects not only the contralateral M1-S1 but also the ipsilateral M1-S1
would subserve the preparation and execution of volitional unilateral one-digit movements.
It can be speculated that EEG, MEG, and fMR integration could be largely useful for a
study with maximum spatial-temporal resolution of dynamic cortical responses in normal and
diseased subjects.
References
1. Boecker H, Kleinschmidt A, and Requardt M, Functional cooperativity
of human cortical motor areas during self-paced simple finger movements: a high resolution
MR study, Brain, 117 (1994) 1231-1239
2. Chollet F, Di Piero V, Wise RJS, Brooks DJ, Dolan R, and Frackowiak
RSJ, The functional anatomy of motor recovery after stroke: a study with PET, Ann.
Neurol., 23 (1991) 63-71
3. Colebacht JG, Deiber MP, Passingham RE, Friston KJ and Frackowiak
RSJ, Regional cerebral blood flow during voluntary arm and hand movements in human
subjects, J. Neurophysiol., 65-6 (1991) 1392-1401
4. Fuchs M, Wagner M, Wischmann H-A, Kohler T, Theissen A et al,
Improving source reconstruction by combining bioelectrical and biomagnetic data Electroenceph.
clin. Neurophysiol., 107 (1998), 2, 69-80.
5. Haaland K, and Delaney HD, Motor deficits after left or right
hemisphere damage due to stroke or tumor, Neuropsychol., 19 (1981) 17-27.
6. Haaland K, and Harrington D, Hemispherical control of the initial
and corrective components of aiming movements, Neuropsychol., 27 (1989) 961-969.
7. Hämäläinen M, and Sarvas J, 1989 Realistic conductivity geometry
model of the human head for interpretation of neuromagnetic data IEEE Trans. Biomed.
Eng., 36 165-171
8. Hansen PC 1992 Analysis of discrete ill-posed problems by means of
the L-curve SIAM Review, 34 561-580
9. Jones RD, Donaldson IM, and Parkin PJ, Impairment and recovery of
ipsilateral sensory-motor function following unilateral cerebral infarction, Brain, 112
(1989) 113-132
10. Grave de Peralta R, Hauk O, Gonzalez Andino S, Vogt H, and Michel
CM, Linear inverse solution with optimal resolution kernels applied to the electromagnetic
tomography, Human Brain Mapping, vol. 5., pp. 454-467, 1997.
11. Kim SG, Ashe J, Georgopoulos AP, Merkle H, Ellermann JM, Menon RS,
Ogawa S, and Ugurbil K, Functional imaging of human motor cortex at high magnetic field,
J. Neurophysiol., 69 (1993a) 297-302.
12. Kim S, Ashe J, Hendrich K, Ellermann J, Merkle H, Ugurbil K, and
Georgopulos A, Functional magnetic resonance imaging of motor cortex: hemispherical
asymmetry and handedness, Science, 261 (1993b) 615-617.
13. Kristeva R, Cheyne D, and Deecke L, 1991 Neuromagnetic fields
accompanying unilateral and bilateral voluntary movements: topography and analysis of
cortical sources Electroenceph. clin. Neurophysiol. 81 284-298
14. Meyer BU, Roericht S, Graefin von Einsiedel H, Kruggel F, and
Weindl A, Inibitory and excitatory interhemispheric transfers between motor cortical areas
in normal humans and patients with abnormalities of the corpus callosum, Brain, 118
(1995) 429-440
15. Meijs J, Weier O, Peters M, and van Oosterom A, 1993 On the
numerical accuracy of the boundary element method IEEE Trans. Biomed. Eng., 42,
1038-1049
16. Oldfield P, The Edimburgh Inventory, Neuropsychol. 3 (1971)
124-132.
17. Rao SM, Binder JR, Bandettini PA, Hammeke TA, Yetkin FZ,
Jesmanowitcz A, List LM, Morris GL, Mueller WM, et al., Functional magnetic resonance
imaging of complex human movements, Neurol., 43 (1993) 2311-2318.
18. Rao SM, Binder JR, Hammeke TA, Bandettini PA, Bobholz JA, Frost JA,
Myklebust M et al., Somatotopic mapping of the human primary motor cortex with functional
magnetic resonance imaging, Neurol., 45 (1995) 919-924.
19. Roland PE, Larsen B, Lassen NA, and Skinohoj E, SMA and other
cortical areas in organization of voluntary movements in man, J. Neurophysiol., 43 (1980)
118-136.
20. Rouiller E, Babalian A, Kazennikov V, Moret V, Yu X, and
Wiesendanger M, Transcallosal connections of the distal forelimb representations of the
primary and supplementary motor cortical areas in macaque monkeys, Exp. Brain Res., 102
(1994) 227-243.
21. Sadato N, Yonekura Y, Waki A, Yamada H, and Ishii Y, Role of the
SMA and the right premotor cortex in the coordination of bimanual finger movements, J.
Neurosci., 17-24 (1997) 9667-9674
22. Shibasaki H, Sadato N, Lyshokow H, Yonekura M, Nagamine T, et al.,
Both primary motor cortex and SMA play an important role in complex finger movement,
Brain, 11 (1993) 1387-1398
23. Scheibel AB, The brainstem reticular core and sensory function. In:
Darian-Smith, I. (Ed.), Handbook of Physiology, The Nervous System. American Physiology
Society, Bethesda, 1984, pp. 213-256.
24. Schroeder J, Wenz F, Schad LR, Baudendistel K, Knopp MV,
Sensorimotor cortex and SMA changes in schizophrenia, A study with functional magnetic
resonance imaging, J. Psych., 167 (1995) 197-201
25.Tanji J, Ohano K, and Sato K, Neuronal activity in cortical motor
areas related to ipsilateral, contralateral, and bilateral digit movements of the monkey,
J. Neurophysiol., 60 (1988) 325-343
26. Urbano A, Babiloni C, Onorati P, and Babiloni F, 1996 Human
cortical activity related to unilateral movements. A high resolution EEG study NeuroReport,
8, 203-206
27. Wassermann EM, Pascual-Leone A, and Hallett M, Cortical motor
representation of the ipsilateral hand and arm, Exp. Brain Res., 100 (1994) 121-132
28. Weiller C, Juptner M, Fellows S, Rijntjes M, Leonhardt G, Kiebel S,
Muller S, Diener HC, Thilmann AF, Brain representation of active and passive movements,
Neuroimage, 4 (1996) 105-110
29. Wiesendanger M, Rouiller E, Kazelnnikov O, and Perrig S, Is the SMA
a bilaterally organized system? Adv. Neurol., 70 (1996) 71-83.
|

 Home
Current Issue
Table of Contents
Home
Current Issue
Table of Contents

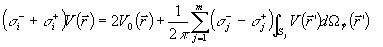
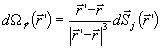
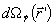 is the solid angle subtended by the surface element dS located in
is the solid angle subtended by the surface element dS located in
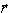 (point of observation
(point of observation