|
Utilization of MRI Information in EEG Studies
Hannu Eskola
Ragnar Granit Institute, Tampere University of Technology, P.O.Box 692, 33101 Tampere, Finland
Abstract.This article introduces various methods to combine MR images and EEG signals. The methods are presented in
historical order, beginning from so-called "brain mapping" in 1980´s and ending up with potential
applications in the future. The motivation for this kind of review is in the conviction that the combination of
these two modalities is strongly increasing today, due to important steps in the development of both methods.
1. Introduction
The advances in the clinical application of the electroencephalography (EEG) have always been connected with
technical innovations, such as the string galvanometer, transistor and integrated circuit (IC) technology. These
have enabled sufficient signal to noise ratio as well as small size and low prize of the multi-channel electronics.
In the beginning of 1980´s it was understood that the development of the computer technology would create
a new jump in the technology of EEG, which was realized by the fast breakthrough of digital EEG during this decade.
The next decade may provide us with a new imaging technology, EEG.
Since about 1980 it has been possible to record EEG digitally. From the very beginning this was applied to the
approximation of topographic maps of scalp potentials [1]. In many respects, however, the technological circumstances
did not meet the requirements of the users. The first devices consisted of 16 channels, screen and printer resolution
of about 200 lines, and produced samples of only eight bits. Additionally, the quality of the raw signal was often
worse than that produced by the conventional devices. The consequence was that the topographic maps resulting from
the first digital EEG devices did not convince the EEG experts. This period may have influenced the slow progress
in application of the local information as a part of EEG diagnostics.
In 1990´s digital EEG devices followed the EEG mapping computers. These provide the users
typically with 32 channels and 16 bits, ensuring that nothing is lost from the original analogue information. Despite
of the remarkable changes in EEG interpretation practice, the neurophysiologists have adapted the new paperless
methods. Although there is some reluctance to the digital EEG protocols, there are no EEG laboratories in USA anymore
who are going to purchase paper EEG devices [2].
The rapid digitalization of EEG laboratories gives various options for the users to process the data. One of
these is the combination of the data with MRI information. This is a relevant option today, because many neurological
patients undergo diagnostic MRI examinations. The combination of these two modalities can be performed in various
ways, as will be shown in the next chapter. Some of these methods can be found in commercial devices, some are
under development in research laboratories, whereas some are still hypothetical.
2. Methods for combining MRI and EEG information
There are several levels for the utilization of MRI information in EEG studies. By increasing the complexity
level more specific information is obtained. The complexity involves processing of both the EEG and MRI data. Various
processing methods are described in Figure 1, as well as the resulting combinations.
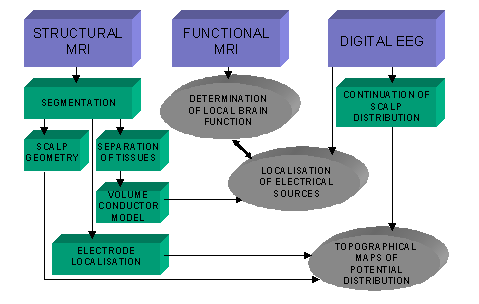
Figure 1. Procedures for combining the information from EEG signals and MR images.
2.1 Illustration of scalp EEG maps
Topographical map is one way to present EEG information, an alternative for the EEG curves. The potentials at
a certain moment or period of time are coded as colors at the corresponding electrode locations. The electrical
potential values on spaces between the electrode points are approximated by using, for example, interpolation or
spline fitting methods. For this purpose, the scalp must somehow be shown as a 3D image on the screen or as a printout.
The simplest way to present the scalp is a general sketch (spherical or elliptical) of the head as a cranial
projection. In some mapping realizations there are also anterior, posterior and lateral views. To enable more realistic
illustration, these views can be obtained from optical or MRI images of a typical human subject. This is probably
the simplest way to combine EEG and MRI data.
>
A more sophisticated method is based on individual anatomy of the scalp, which is obtainable from segmented
MRI data [3, 4, 5]. If the EEG lead system is based on anatomical landmarks (as is the case in standard 10-20 and
10-10 systems), the EEG electrode locations may be approximated from the MRI images. This can be done by searching
the anatomical landmarks (for instance ears, nasion and inion) from the image and using the geometrical rules of
the lead system for calculating the theoretical positions of the electrodes [6]
As the MRI image is available, the real locations of EEG electrodes are often desired. Numerous methods have
been used for this purpose. The electrodes can be marked for instance by touching the electrode locations by a
3D scanner, which operates in a magnetic field. These kind of methods produce an optical image of the head, which
must be matched with the MRI image [3]. The matching can be avoided if MRI positive markers are placed at the electrode
locations after the EEG study. This method involves that the MRI imaging can be performed quite soon after the
EEG recording [7]. Figure 2 shows a typical result of this kind of map.

Figure 2. A topographical map presentation of the EEG potentials. The anatomy
and the markers for the electrode locations of the subject are segmented from a T1 MRI slice set.
The potential distribution on the scalp can be processed in many ways. A continuation procedure is always necessary
to transform the discrete voltage values into a continuous potential distribution. Interpolation of the values
gives a rough estimate, resulting in piece-vice linear areas. More realistic estimates constitute of fits to continuous
two-dimensional distributions, such as 2D splines.
The original potential values can also be altered in order to increase the spatial information. One of the most
promising methods is the surface Laplacian algorithm [9]. It produces an estimate of the radial current at each
electrode point. This estimate is based on a comparison of the potential value to those of the nearest neighboring
electrodes or, in optimal case, of all other electrodes. The accuracy depends also on the model for the head shape
[4].
These advanced methods for the evaluation of the scalp EEG maps can quite effectively make use of the anatomical
information obtained from MRI scanning. They also show that more spatial information may be obtained from EEG by
increasing the number of electrodes from the standard 10-20 system. Figure 3 shows the effect of increase of recording
points from 21 to 64. The amount of details in EEG maps still increases as 128 electrodes are used [10]. This corresponds
to decreasing the inter-electrode distance from about 7 cm to 3 cm. An essential observation is that the ordinary
potential maps do not change dramatically. However, by applying the Laplacian maps the spatial information increases
with the number of electrodes [4].
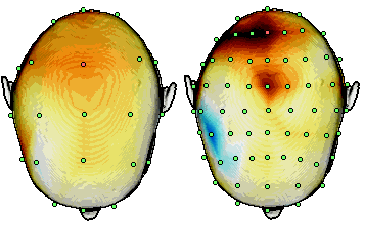
Figure 3. A topographical map presentation of EEG Laplacians during drowsiness recorded by
using 21 (a) and 64 (b) channels. The anatomy of the subject and the electrode locations have been
segmented from a T1 MRI slice set. Modified from Manninen et al. [11].
2.2 Reconstruction of intracranial surface maps
The scalp potential distribution reflects the distribution of intracranial electrical sources and fields. It
can obviously be used for the approximation of the cortical potential distribution. Spatial deconvolution methods
such as the finite element deblurring method [12] have been presented and validated by using both cortical recordings
and computational models [13, 14]. The potentials are not usually evaluated on the complicated cortical surface
itself, but on a more regular surface locating few millimeters apart from the cortex. Therefore the distributions
are sometimes called intracranial or dural maps.
Realistic intracranial maps involve the solution of the inverse problem. This means that the maps are not unique
and that the calculation of the maps takes a considerable amount of time. Unlike Laplacians and other cortical
maps, the intracranial maps cannot be computed in real time. More pre-processing is also required, such as the
segmentation of the scalp and skull for the potential estimation and the segmentation of the cortical surface for
displaying the map in 3D. The procedure is considered in more detail in next section.
2.3 Localization of the source activity
The electrophysiology of the brain is very complicated. The EEG is roughly divided into event-related potentials
and spontaneous activity. Both are distributed in a wide range of the brain, but especially some periods of event-related
potentials are focused in certain locations. Also certain abnormal activity, such as epileptic spikes, may originate
focally in the brain. These cases fulfill the conditions of the traditional source analysis. It is based on the
assumption that the EEG can be described by one electrical point dipole source at a certain moment or period of
time. More advanced methods imply multiple dipoles and distributed sources, which are in most cases more realistic
models for the electrical brain activity [15]
Source analysis implies the solution of the inverse problem, which is based on several solutions of the forward
problem. Not only the source has to be modeled, but also the head must be replaced by a volume conductor model
/Malmivuo et al./. This model is in all practical solutions linear, resistive, piece-vice homogeneous and isotropic.
In principle it would be possible to deal with even more realistic models, which have anisotropic and complex impedances.
Further, the minimum size of each compartment in the model could be the dimension of the voxels obtained from the
MRI scan. However, all these features demand lot of computing power and are therefore not included in present head
models.
The forward models of the head can be divided into spherical [17, 18] and anatomical [19 - 23] realizations.
Spherical models work fast and are thus optimal for patient studies consisting of large materials. Anatomical models
are time-consuming, but give the individual geometry of the subject and suit for special cases.
Although MRI is not needed for the computation in the former case, the MRI scans are often used also in spherical
modeling. The sphere can be positioned either in the center of the head or asymmetrically so as to produce an optimal
source localization. [24]. Independently on the modeling method, the MRI images are often used for displaying the
anatomical locations of the sources.
2.4 Evaluation of the electrical field in the brain
As the electrical sources in the brain have been estimated, it is possible to use the forward approach to solve
the electrical field everywhere in the brain. However, the intracranial electrical field distribution seems not
to have direct relevance in clinical or neuropsychological applications. The method has mostly been applied in
visualization of simulation studies, i.e., in forward problems. In association with the inverse problem it involves
heavy computation and has not been studied extensively.
On the other hand, single point dipole gives hardly a realistic representation of the whole electrical activity.
For this reason special algorithms have been developed which enable the estimation of the electrical activity overall
in the brain without performing the actual source localization procedure [25]. These methods are based on a priori
knowledge on the spread of the activation in the neural tissues.
2.5 Temporal behavior of intracranial electrical activity
The power of EEG is mostly in its properties in temporal space. The temporal resolution itself is one or two
orders of magnitude better than in the imaging methods. EEG is also basically considered as a signal, not an image,
and most of the interpretation is based on the waveform analysis. Therefore it would be useful to include the temporal
dimension to all analysis methods described in earlier sections.
Adding the time dimension to topographic potential maps produces a movie option, where the rate of change of
the maps can be adjusted in a wide range around the real speed. This option has found to be useful in all kind
of analysis of the digital EEG. In principle it is also possible to follow the movements and rotations of single
or multiple dipoles in the brain. However, the speed of this procedure is reasonable only if the model is properly
simplified.
2.6 Co-registration of EEG and MRI
New inventions in EEG technology have enabled the recording of EEG while the patient is positioned in the MRI
magnet tube, even during the MRI recording. For this purpose several problems have been overcome. MRI imaging uses
RF pulses for exciting the tissue, inducing also interference peaks in the EEG recording. On the other hand, metallic
EEG electrodes and especially the wires affect the magnetic fields and blur certain parts of the image. These disturbances
are attenuated when special instrumentation and methodologies are used [26].
Several methods have been suggested for applying the EEG in MRI room. Epileptic spikes in EEG may be used for
triggering the functional MRI session. Also successful co-registration of EEG and MRI has been reported with acceptable
quality of EEG with the exception of the period during the RF pulses.
3. Organization of a multimodal EEG protocol
There are few research laboratories that have both the MRI and EEG instrumentation. To combine
these studies, co-operation with the hospitals is usually essential. Medical projects involve also close collaboration
between the clinicians and the research personnel. Figure 4 shows a result of such collaboration between several
departments of Tampere University Hospital, Faculty of Medicine in University of Tampere, and Ragnar Granit Institute
in Tampere University of Technology. The video EEG recordings, MRI scans, spike detection and EEG interpretation,
combination of EEG and MRI, 3D representation of the results, as well as the meeting for the clinical conclusions
were all scheduled into one week.
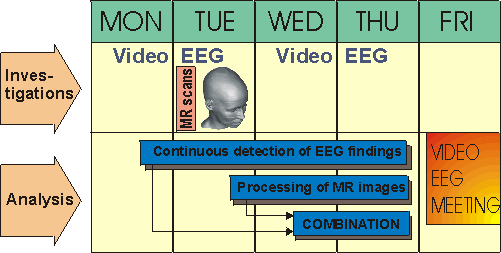
Figure 4. Weekly schedule for a selected epileptic patient, including video EEG and MRI studies
and processing of the data.
Scheduling is only one part of organizing multimodal EEG+MRI studies. Several specialists are needed who all
should be research orientated and motivated to the project:
- neurologists
- clinical neurophysiologists (EEG experts)
- EEG technicians
- EEG engineers or physicists
- radiologists
- radiographers
- MRI physicists
- researchers in physics/engineering
- researchers in medicine
A general conclusion from these requirements is that whenever several investigations are combined, the organizational
challenges increase faster than the amount of individual examinations. This sets high requirements for the management
of any project involving the combination of EEG and MRI studies.
References
[1] Duffy F, Denckla M, Bartels P, Sandini G, Dyslexia: regional differences in brain electrical activity by topographic
mapping. Ann Neurol 7(5):412-20, 1980.
[2] Brenner R, Scheuer M, Cross-country digital EEG survey 1998. J Clin Neurophysiol 15(6):485-8, 1998.
[3] Gevins A, Brickett P, Costales B, Le J, Reutter B, Beyond topographic mapping: towards functional-anatomical
imaging with 124-channel EEGs and 3-D MRIs. Brain Topogr 3(1):53-64, 1990.
[4] Babiloni F, Babiloni C, Carducci F, Fattorini L, Onorati P, Urbano A, Spline Laplacian estimate of EEG potentials
over a realistic magnetic resonance-constructed scalp surface model. Electroencephalogr Clin Neurophysiol 98(4):363-73,1996.
[5] Heinonen T, Eskola H, Dastidar P, Laarne P, Malmivuo J: Segmentation of T1 MR scans reconstruction of resistive
head models, Computer Methods and Programs in Biomedicine 54:173-181, 1997.
[6] Heinonen T, Lahtinen A, Häkkinen V, Implementation of Three-Dimensional EEG Brain mapping, Computers and
Biomedical Research, in press, 1999.
[7] Laarne P, Solution of the Electric Forward Problem Using a Realistic Head Model. Licentiate thesis, Tampere
University of Technology, 1996.
[8] Babiloni F, Babiloni C, Fattorini L, Carducci F, Onorati P, Urbano A, Performances of surface Laplacian estimators:
a study of simulated and real scalp potential distributions. Brain Topogr 8(1):35-45, 1995.
[9] Nunez P, Estimation of large scale neocortical source activity with EEG surface Laplacians. Brain Topogr, 2(1-2):141-54,
1989.
[10] Srinivasan R, Tucker D, Murias M, Estimating the spatial Nyquist of the human EEG. Behavior Research Methods,
Instruments and Computers 30(1), 8-19, 1998.
[11] Manninen M, Hirvonen K, Lahtinen A, Malmivuo J, Eskola H, Visualization of topographical
changes in EEG during drowsiness and sleep onset . Accepted to: Med Biol Eng Comput, 1999. 158
[12] Gevins A, Le J, Brickett P, Reutter B, Desmond J, Seeing through the skull: advanced EEGs use MRIs to accurately
measure cortical activity from the scalp. Brain Topogr 4(2):125-31, 1991.
[13] Gevins A, Le J, Martin N, Brickett P, Desmond J, Reutter B, High resolution EEG: 124-channel recording, spatial
deblurring and MRI integration methods. Electroencephalogr Clin Neurophysiol 90(5):337-58, 1994.
[14] Nunez P, Silberstein R, Cadusch P, Wijesinghe R, Westdorp A, Srinivasan R, A theoretical and experimental study
of high resolution EEG based on surface Laplacians and cortical imaging. Electroencephalogr Clin Neurophysiol 90(1):40-57,
1994.
[15] Mosher JC, Leahy RM, Recursive MUSIC: a framework for EEG and MEG source localization. IEEE Trans Biomed Eng
45(11):1342-54, 1998.
[16] Malmivuo J, Plonsey R, Bioelectromagnetism - Principles and Applications of Bioelectric and Biomagnetic Fields.
Oxford University Press, New York, 1995.
[17] Brazier M, A study of the electric field at the surface of the head. Electroencephalogr Clin Neurophysiol 2(Suppl
1):38-52, 1949.
[18] Berg P, Scherg M, A fast method for forward computation of multiple-shell spherical head models. Electroencephalogr
Clin Neurophysiol 90(1):58-64, 1994.
[19] Laarne P, Eskola H, Hyttinen J, Suihko V, Malmivuo J, Validation of a detailed computer model for the electric
fields in the brain. J Med Eng Technol 19(2-3):84-7, 1995.
[20] Zanow F, Peters MJ, Individually shaped volume conductor models of the head in EEG source localisation. Med
Biol Eng Comput; 33(4):582-8, 1995.
[21] Lemieux L, McBride A, Hand J, Calculation of electrical potentials on the surface of a realistic head model
by finite differences. Phys Med Biol 41(7):1079-91, 1996.
[22] Haueisen J, Ramon C, Eiselt M, Brauer H, Nowak H, Influence of tissue resistivities on neuromagnetic fields
and electric potentials studied with a finite element model of the head. IEEE Trans Biomed Eng 44(8):727-35,1997.
[23] Fuchs M, Drenckhahn R, Wischmann HA, Wagner M, An improved boundary element method for realistic volume-conductor
modeling. IEEE Trans Biomed Eng 45(8):980-97, 1998.
[24] Towle VL, Bolanos J, Suarez D, Tan K, Grzeszczuk R, Levin DN, Cakmur R, Frank SA, Spire JP, The spatial location
of EEG electrodes: locating the best-fitting sphere relative to cortical anatomy. Electroencephalogr Clin Neurophysiol
86(1):1-6,1993.
[25] Pascual-Marqui RD, Michel CM, Lehmann D, Low resolution electromagnetic tomography: a new method for localizing
electrical activity in the brain. Int J Psychophysiol 18(1):49-65, 1994.
[26] Allen PJ, Polizzi G, Krakow K, Fish DR, Lemieux L, Identification of EEG events in the MR scanner: the problem
of pulse artifact and a method for its subtraction. Neuroimage 8(3):229-39, 1998.
|

 Home
Current Issue
Table of Contents
Home
Current Issue
Table of Contents




