|
Combining the Electrical and Mechanical Functions of the Heart
Frank B Sachse, Gunnar Seemann, Christian D Werner
Institut für Biomedizinische Technik, Universität Karlsruhe
(TH), D 76128 Karlsruhe
Correspondence: Frank B. Sachse, Institut für Biomedizinische
Technik, Universität Karlsruhe (TH),
Kaiserstr. 12, D 76128 Karlsruhe,
Germany,
E-mail: Frank.Sachse@ibt.etec.uni-karlsruhe.de, phone +49 721
608 3851, fax +49 721 608 2789<
Abstract.
Computer aided simulations of the heart provide knowledge for cardiologic
diagnosis and therapy. A model of the myocardium is presented which allows
the reconstruction of electrical and mechanical processes with inclusion
of feedback mechanisms. The model combines detailed models of cellular
electrophysiology and force development with models of the electrical current
flow and the mechanical behavior of the myocardium. Results of simulations
show the connection between the electrical excitation process and the following
mechanical deformation in a three dimensional, anisotropic area of the
myocardium.
Keywords: Mechano-Electrical Feedback, Electro-Mechanical Feedback,
Cellular Models, Electrophysiology, Excitation-Propagation
1. Introduction
The understanding of the physiological and pathophysiological behavior
of the heart can be improved by computer aided simulations of the electrical
and mechanical processes in the myocardium. These simulations are of importance
e.g. for studying cardiac arrhythmias and the influence of medicaments
as well as for the computer aided planning of surgical interventions. Different
approaches allow the simulation of electromechanical processes in the myocardium.
Commonly, the approaches are restricted to a reconstruction of either electrophysiological
or mechanical processes. Hereby, the influence of feedback mechanisms is
neglected. The approach presented in this work consists of combining electrophysiological
models of single myocardial cells with models of the electrical current
flow through the myocardium. The electrophysiological models are coupled
with models of the cellular force development. The calculated forces are
used by an elastomechanical model of the myocardium. The resulting deformation
is delivered to all models e.g. to consider feedback mechanisms. The approach
is used to generate a three dimensional model of an area of the ventricular
myocardium. The model allows the simulation of the electrical excitation
propagation, force development and deformation. The model is derived from
a detailed electrophysiological model of ventricular cells [ Noble
et al., 1998]. The electrical coupling of cells is achieved using of
an extension of the bidomain model, which consists of calculation of the
intracellular and extracellular current flow [ Sachse
et al., 2000]. Therefore, Poisson's equation for electrical current
fields is applied on grids, which are geometrically deformed by the stretch.
The force model described in [ Rice et al., 1999]
uses parameters delivered by the electrophysiological model. The elastomechanical
model is generated from a strain energy function proposed by [ Hunter
et al., 1998] and numerical methods of continuum mechanics [ Bathe,
1996]. The calculation takes the orientation and lamination of myocytes
into account, which leads to an anisotropy of the electrical conductivity
and the elastomechanical behavior.
2. Methods
2.1 Microscopic Anatomy of the
Human Heart
2.1.1 Overview
The myocard consists primarily of discrete myocytes, which are arranged
in an oriented and laminated structure [ Streeter
and Bassett, 1966, Streeter, 1979, LeGrice
et al., 1995]. The myocytes are surrounded by the extracellular space,
which contains e.g. collagen fibers, water, and electrolytes. Additionally,
the myocard is pervaded by nerves, capillaries, blood vessels, and lymphatic
vessels. It can be distinguished between myocytes assigned to the working
myocard and myocytes with the task of initiating and transmitting electrical
excitation. This work is focussed upon the myocytes of the working myocard.
2.1.2 Myocytes of the Working
Myocard
Myocytes are enclosed by the sarcolemma, a phospholipid bilayer, which delimits
the extracellular from the intracellular space. In the intracellular
space resides the nucleus, the mitochondria, and the myofibrils.
Myofibrils are tube shaped contractile elements taking a high
part of the cellular volume. They have a thickness of approximately 1 μm
and are divided every approximately 2.5 μm
by the Z disk into the sarcomeres. The sarcomere contains
the myofilaments, which are of importance for the mechanical
contraction. The myofilaments are composed of actin (thin)
and myosin (thick) filaments [ Bers, 1991].
The thin filaments lead from the Z disks approximately 1 μm
towards the center of the sarcomere. The filaments consist
of long, flexible, coiled coil molecules, i.e. tropomyosin,
at which the actin and troponin is attached. At every seventh
actin a troponin is bound to the tropomyosin. Troponin consists
of three components: troponin T, troponin I and troponin C.
Troponin T is connected to the tropomyosin and the two further
components. Troponin I inhibits interactions between actin
and myosin. Troponin C is of importance for the initiation
of the mechanical contraction by binding of calcium. The thick
filaments have a length of approximately 1.6 μm
and are centered in the sarcomere. They are parallel to and
arranged between the thin filaments. The thick filament is
composed primarily of the myosin molecule, which has a length
of approximately 160 nm.
Myosin in myocytes of cardiac, skeletal and smooth muscles
is subdivided into two heads and tails. The head shows an
ATP and an actin binding site. The tails are coiled coil molecules
and form the main axis of the thick filaments. The sarcolemma
intrudes at the adjacencies of the Z disks to form the transversal
tubuli and to infold the myofibrils. The system of longitudinal
tubuli plays in contrast to the skeletal myocytes in cardiomyocytes
only a subsidiary role [ Bers, 1991].
Myocytes are of irregular shape, but a dominant principal
axis can be assigned. This leads to a macroscopic anisotropic
electrical and elastomechanical behavior. The ratios of volume
and area of the cell components are differing for atrial and
ventricular myocytes [ Bers, 1991].
2.1.3 Gap Junctions
The intracellular space of myocytes is coupled by gap junctions (nexus), which
are located by bundle at the intercalated disks [ Dhein,
1998, Dellmar et al., 1999]. Intercalated
disks are disk shaped segments, which mechanically couple
cells [ Forbes and Sperelakis, 1985].
Primarily, the disks can be found at or near to the ends of
myocytes [ Saffitz and Yamada, 1999].
A gap junction is cylinder or barrel shaped with a diameter
of 1.5 - 2.0 nm and a length of approximately 2 - 12 nm.
A molecule of atomic weight up to 1 kD can pass through the
gap junction [ Jongsma and Rook, 1999, Yeager,
1999], e.g. nutrients, metabolites, and ions. A gap junction
is built up by two hemi-channels, so-called connexons, piercing
the sarcolemma of the involved cells. The connexons are formed
by six integral membrane proteins, so-called connexins. More
than one dozen of connexins have been cloned, which are named
by their atomic weight ranging from 25 to 50 kD. The most
abundant connexin in the mammalian myocard is connexin43,
which is also expressed in ovary, uterus, kidney, and lens
epithelium. The connexin32 is found in liver, stomach, kidney,
and brain, but not in any part of the heart. In the myocard
each myocyte is coupled by gap junctions non-uniformly with
other myocytes, e.g. in canine a myocytes is coupled with 9.1 ± 2.2
myocytes [ Hoyt et al., 1989]. A distinction
can be made between longitudinal and transversal gap junctions.
A longitudinal gap junction is oriented in approximately the
same direction as the first principal axis of adjacent myocytes,
a transversal gap junction is oriented perpendicular thereto.
The density and distribution of orientations of gap junctions
differs depending on the tissue, e.g. the density in the sinus
and atrioventricular node is smaller than in the ventricular
myocard. The average density of gap junctions in longitudinal
orientation is larger than the transversal orientation [ Hoyt
et al., 1989]. The average length of longitudinal gap
junctions is smaller than the length of transversal gap junctions.
Both circumstances lead to a macroscopic anisotropic electrical
conductivity.
2.1.4 Cardiac Collagen Network
The myocard is pervaded and surrounded by a mesh of extracellular collagen fibers,
which are composed of a multitude of collagen fibrils synthesized
by the cardiac fibrocytes. Collagen takes 2 - 5 %
of the weight of the heart. Furthermore, fibers of elastin
draw through the myocard. The content and the structure of
the connective tissue are dependent on age, pathologies and
species. Both, collagen and elastin are fibrous proteins of
the extracellular matrix, which is a determinant for the viscoelastic
behavior of the myocard. The network serves for the mechanical
coupling of the myocytes, capillaries, and lymphatic vessels.
Collagen fibrils have a thickness from 30 - 70 nm
[ Caulfield and Borg, 1979]. The
fiber thickness is in physiologic cases between 120 and 150 nm.
An increase up to 250 - 300 nm
is possible in pathophysiologic cases, e.g. hypertrophy, hypertension
and myocardial infarction [ Abrahams
et al., 1987, Ju and Dixon, 1996].
The density of collagen fibers is depending on the tissue.
E.g. a small density can be found in papillary muscle and
trabeculae, a high density in the left ventricular myocard,
the endocardium and epicardium [ Caulfield
and Borg, 1979, Weber et al., 1994].
2.2 Modeling of Cellular Electrophysiology
Table 1: Electrophysiological models of cardiac cells.
| Cell Type |
Species |
Reference |
| Purkinje fiber |
- |
[Noble, 1962] |
| Purkinje fiber |
- |
[McAllister et al., 1975] |
| Ventricular myocard |
mammal |
[Beeler and Reuter, 1977] |
| Purkinje fiber |
mammal |
[DiFrancesco and Noble, 1985] |
| Atrial myocard |
rabbit |
[Hilgemann and Noble, 1987] |
| Atrial myocard |
rabbit |
[Earm and Noble, 1990] |
| Ventricular myocard |
mammal |
[Luo and Rudy, 1991] |
| Ventricular myocard |
guinea-pig |
[Luo and Rudy, 1994a,Luo and Rudy, 1994b] |
| Sinus node |
mammal |
[Demir et al., 1994] |
| Ventricular myocard |
canine |
[Demir et al., 1996] |
| Atrial myocard |
human |
[Courtemanche et al., 1998] |
| Ventricular myocard |
guinea-pig |
[Jafri et al., 1998] |
| Ventricular myocard |
guinea-pig |
[Noble et al., 1998] |
| Atrial myocard |
human |
[Nygren et al., 1998] |
| Ventricular myocard |
human |
[Priebe and Beuckelmann, 1998] |
| Ventricular myocard |
canine |
[Winslow et al., 1999,O'Rourke et al., 1999] |
2.2.1 Overview
The electrophysiological state of cells can be described by a spatial distribution
of ion concentrations, which are changed by passive and active transport
mechanisms. The transport of ions is time dependent as well as influenced
by gradients of concentrations and the electrical field. Most electrophysiological
models of cells are constructed from the classical work of Hodgkin and
Huxley, who described quantitatively the active and passive behavior of
a neuron membrane [ Hodgkin and Huxley, 1952].
Hodgkin and Huxley used voltage clamp techniques to measure currents through
the membrane of giant axons of squids. From this data an equivalent circuit
consisting of resistors, a capacity, and voltage sources was parameterized.
Partly, the resistors were nonlinear time and voltage dependent. In the
last years a large number of models of cardiac cells were constructed (table
1),
with increasing abilities to describe the different electrophysiological
mechanisms. Primarily, the models are produced from animal experiments.
Modern models include detailed descriptions of the behavior of intracellular
structures as well as of the influence of drugs and deformation.
2.2.2 Noble-Varghese-Kohl-Noble
Model
A foundation of this work is the Noble-Varghese-Kohl-Noble model of a ventricular
cell [ Noble et al., 1998]. Thereby,
the time derivative of the transmembrane voltage Vm
is described by:
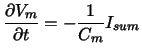 |
(1) |
with the membrane capacity Cm
and the summary current through the membrane Isum
with the following parameters:
| background K current |
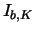 |
| time-independent K current |
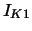 |
| time-dependent, delayed K current |
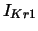 , , 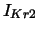 , , 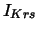 |
| ACh-dependent K current |
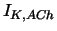 |
| background Na current |
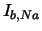 |
| fast Na current |
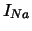 |
| voltage dependent Na current |
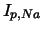 |
| background Ca current |
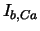 |
| L-type Ca current |
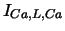 , ,
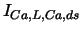 , ,
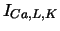 , , |
| |
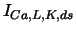 , ,
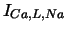 , ,
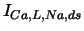 |
| Na-K exchange pump current |
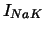 |
| Na-Ca exchange pump current |
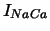 , ,
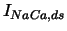 |
| stretch activated current |
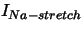 , ,
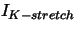 , ,
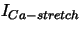 , , |
| |
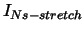 , ,
 |
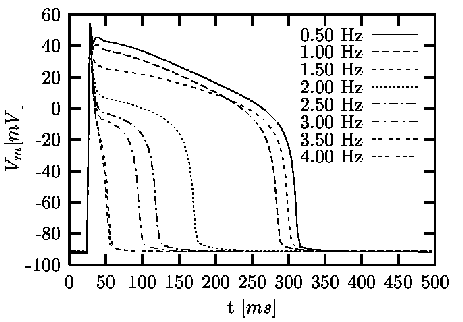
Figure 1: Simulations with the Noble-Varghese-Kohl-Noble
model. The transmembrane voltage Vm
is dependent on the stimulus frequency. For each frequency
a single action potential is visualized [Sachse,
2001].
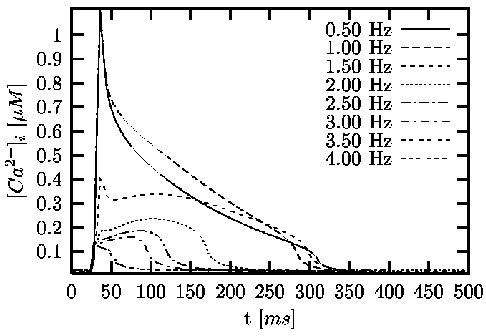
Figure 2: Simulations with the Noble-Varghese-Kohl-Noble
model. The intracellular calcium concentration [Ca2+]i
is dependent on the stimulus frequency. For each frequency
a single course of the calcium concentration is visualized
[Sachse, 2001].
The figures 1 and 2
shows the influence of stimulus frequency to the course of
the transmembrane voltage Vm
and intracellular calcium concentration [Ca2+]i.
Thereby, stretch activated currents are neglected. With higher
stimulus frequency the resting voltage is increased and the
duration of the action voltage is decreased.
2.2.3 Intracellular Mechano-Electrical
Feedback
The Noble-Varghese-Kohl-Noble model includes dependencies of electrophysiological
parameters on the length or tension of the sarcomere. The mechano-electrical
feedback is realized by introducing selective and non selective stretch-activated
ion conductances, a length/tension dependent modulation of calcium binding
to troponin and a length/tension dependent modulated sarcoplasmatic leak
current. A modification of the model is performed, whereby an adaption
according to measurements published in [ White et al.,
1993] is obtained [ Sachse et al., 2000]. This
modification concerns the stretch dependent action potential duration.
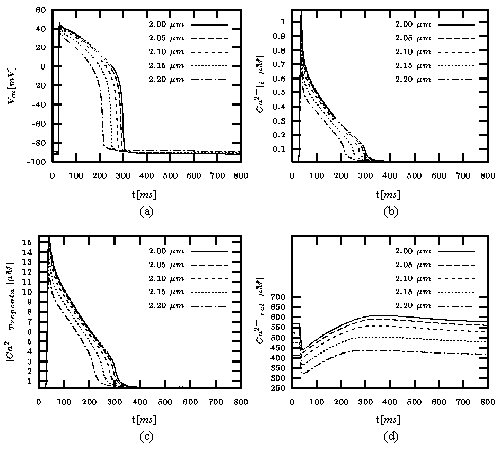
Figure 3: (a) Transmembrane
voltage, (b) calcium concentration in the cytoplasm, (c)
concentration of calcium bound to troponin C, and (d)
concentration of calcium in the release part of the sarcoplasmic
reticulum dependent on length of sarcomere calculated
with Noble-Varghese-Kohl-Noble model (from [Sachse,
2001]). The cell is excited by applying a stimulus
current at t = 25 ms
with a length of 3 ms.
The sarcomere length ranges from 2.0
to 2.2 μm.
The default length of the sarcomere is 2.2 μm.
The stimulus frequency was set to 1 Hz.
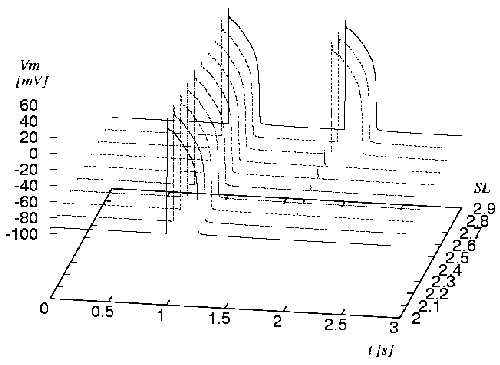
Figure 4: Initiation of
action impulse by stretch of sarcomere. The cell is excited
by applying a stimulus current at t = 1 s.
At t = 2 s
a mechanical stretch of 5 ms
was performed delivering a sarcomere length SL
from 2 to 2.9 μm.
The default length of the sarcomere is 2 μm
(from [Sachse et al., 2000]).
The influence of stretch on the run of the transmembrane potential is illustrated
in figure 3. Thereby, the stretch is specified
by the length of the sarcomere with a default of 2 μm.
The resting potential as well as the progression of the action
potential are dependent on the length of the sarcomere. The
initiation of an excitation by mechanical stretch is depicted
in figure 4. The stretch is applied for
a duration of 5 ms
with varying strength. Depending on the strength of stretch
an effect ranging from a small change of the resting potential
to an excitation of the cell can be achieved.
2.3 Modeling of Excitation Propagation
2.3.1 Approaches
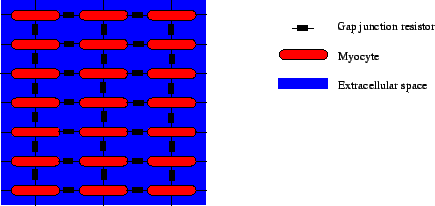
Figure 5: Modeling of electrical intercellular
coupling. The myocytes are coupled via gap junctions and through the extracellular
space.
A propagation of electrical excitation from one cell to neighboring cells
is primarily achieved by intercellular transport of ions via the gap junctions.
Also extracellular potentials resulting from the electrical activity of
cells or from an external current flow can modulate the propagation and
initiate an excitation (figure 5). Two different
classes of approximations of the excitation propagation in the myocard
can be distinguished: microscopic and macroscopic approaches. The macroscopic
approach allows the combining of groups of cells and their common treatment.
In contrast, microscopic models at a cellular level split cells in components,
which are separately treated. In the last years different approaches for
the macroscopic excitation propagation were developed:
-
Cellular automatons. Rules are included defining the time delay and the
neighborhood for the propagation [Eifler and Plonsey,
1975,Killmann et al., 1991,Saxberg
and Cohen, 1991,Wei et al., 1995,Siregar
et al., 1996,Werner et al., 1998,Siregar
et al., 1998].
-
Excitable dynamics equations or reaction diffusion systems
[FitzHugh, 1961,Rogers
and McCulloch, 1994,Panfilov, 1999].
-
Resistor networks/monodomain models. These models incorporate the effect
of coupling the intracellular space with gap junctions [Rudy
and Quan, 1989,Virag et al., 1999].
-
Bidomain models. Bidomain models are an extension of monodomain models
including the effects of the extracellular space [Henriquez
and Plonsey, 1989,Sepulveda and Wikswo, 1994,Henriquez
et al., 1996].
All these models allow the inclusion of anisotropic effects resulting from
the orientation of myocytes, e.g. by using conductivity tensors. Microscopic
models deliver information concerning the stochastic behavior of the myocard
[ Spach and Heidlage, 1995, Spach
et al., 1999]. Anisotropic effects are implicitly included by the cell
geometry as well as by the distribution and orientation of gap junctions.
2.3.2 Bidomain Model
The bidomain model treats the electrical behavior of tissue in two domains,
in the intracellular and extracellular space, which are separated by the
cell membrane. In each domain Poisson's equation for fields of stationary
electrical current is fulfilled:
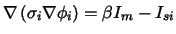 |
|
|
(2) |
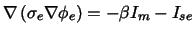 |
|
|
(3) |
with the intracellular potential Φi ,
the extracellular potential Φe,
the intracellular conductivity tensor σi,
the extracellular conductivity tensor σe,
the intracellular current source density Isi,
the extracellular current source density Ise,
and the surface to volume ratio β
of cells. The intracellular conductivity σi
consists of conductivities for the intracellular components
and for the gap junctions. The domains are coupled by the
current density Im
through the cell membrane.
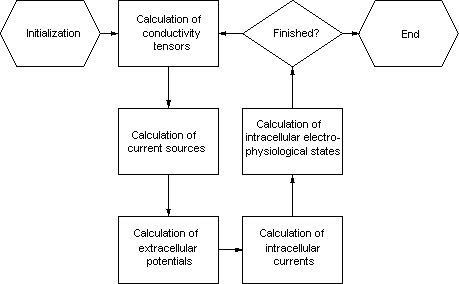
Figure 6: Bidomain modeling of cardiac electrophysiology.
The following method can be chosen to couple the bidomain equations with
the electrophysiological cell models (figure 6)
[Hooke et al., 1992]. The method bases on the iterative
solving of Poisson's equation with numerical techniques:
-
Potentials are determined from current source densities.
-
Current sources are calculated from potentials.
Therefore, commonly the finite-difference or finite-element method is applied
[ Press et al., 1992, Bathe,
1982]. In a first step the current source density Iim
delivered by the transmembrane potential Vm = Φi - Φe
is determined:
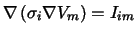 |
|
|
(4) |
In a second step the extracellular potential Φe
is calculated from the current source density Iim:
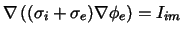 |
|
|
(5) |
The calculation of Φe
is commonly numerically expensive, because the solving of
a large system of linear equations is necessary. In a third
step the intracellular source density Isi
is determined and delivered to the electrophysiological
cell model:
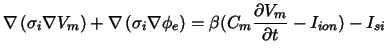 |
|
|
(6) |
The left side of this equation describes a current source density delivered
by the intracellular potentials:
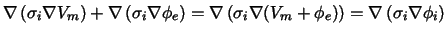 |
|
|
(7) |
2.3.3 Extension of the Mono- and Bidomain Model
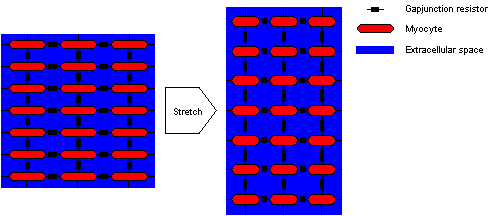
Figure 7: Coupling of myocytes with gap
junctions and through the extracellular space. The deformation of a region
changes the intra- and extracellular conductivity. The resistor yielded
by the gap junction is not changed.
In a previous paper an extension of the mono- and bidomain model was introduced,
which allows to take the deformation of tissue into account
(figure 7) [Sachse
et al., 2000]. This extension delivers conductivity tensors σi
and σe
for the intra- and extracellular space respectively, which
follow the rules of model assumptions. In principal the method
consists of extracting the stretch of regions resulting from
an arbitrary deformation. The extracted stretch is used to
construct a conductivity tensor in a local coordinate system.
Different weights allow to choose a specific behavior of the
conductivity. The local conductivity tensor is transformed
into the global coordinate system.
2.4 Modeling
of Cellular Force Development
2.4.1 Overview
The development of force in the contractile elements of myocytes is provoked by
an increase of the concentration of intracellular calcium [ Ca] i.
The progression of the force is modulated by the progression
of the concentration [ Ca] i.
Commonly, the increase of the concentration [ Ca] i
is a result of an electrical excitation. The progression of
the electrical excitation influences the progression of the
force development (electro-mechanical feedback). Therefore,
many models of cellular force development use the concentration [ Ca] i
to define rate coefficients, which depict the interaction
between states [ Landesberg and Sideman,
1994a, Landesberg and Sideman,
1994b, Rice et al., 1999, Rice
et al., 2000]. The states describe e.g. the binding of
intracellular Ca2+
to the troponin complex and the cross-bridge cycling. Further
parameters influencing the rate coefficients are the sarcomere
length and the state variables.
2.4.2 Rice-Winslow-Hunter Model
Table 2: Tropomyosin and cross bridge states of
Rice-Winslow-Hunter Model 3 of cardiac cells.
| state |
Tropomyosin |
No. of cross bridges |
| N0 |
non permissive |
0 |
| N1 |
non permissive |
1 |
| P0 |
permissive |
0 |
| P1 |
permissive |
1 |
Table 3: Ca2+ binding states of Rice-Winslow-Hunter
Model 3 of cardiac cells.
| state |
Ca2+binding to Troponin C |
| T |
no |
| TCa |
yes |
A foundation of this work are the Rice-Winslow-Hunter models
of cardiac muscle [Rice et al., 1999].
As an example of the modeling a short description of the
3rd model is given. This model consists of 6 states,
N0, N1, P0, P1, T, and TCa
(tables 2 and 3)
with:
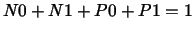 |
|
|
(8) |
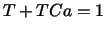 |
|
|
(9) |
The interaction between the states of the model is described by a system
of 1st order differential equations:
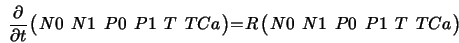 |
(10) |
with the 6 × 6 matrix R
consisting of rate coefficients. Partly, the rate coefficients
are dependent on the sarcomere length SL
and the concentration [Ca]i.
The normalized force F
is determined by
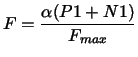 |
|
|
(11) |
with the sarcomere overlap function
α = α(SL)
and the maximal force Fmax.
The states P1 and N1 are the force generating states.
2.5 Modeling of Elastomechanical
Behavior
2.5.1 Principle of Virtual Displacements
The equilibrium of a body is achieved if the internal and external forces are
balanced [ Bathe, 1996]. The equilibrium
at time
t + Δ t
can be expressed using the principle of virtual displacements:
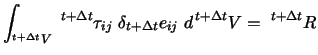 |
(12) |
with the volume t + ΔtV ,
the components of the Cauchy stress tensor t + Δtτij ,
the strain tensor
δt + Δt eij ,
and the external virtual work R.
The formula uses the summation convention of Einstein, where
repeated subscripts become the designation for summation.
The strain tensor is defined as
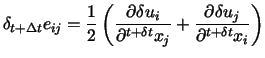 |
(13) |
with the components of the virtual displacement vector δui.
The external virtual work R
is sub-dived in applied force densities t + Δt fiB
and surface tensions t + Δt fiS
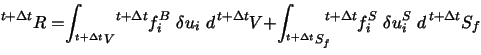 |
(14) |
with the surface
t + Δt S f .
2.5.2 Strain Energy Density Function
The strain energy density function W
proposed by [ Hunter et al., 1997, Hunter
et al., 1998] takes the anisotropic and inhomogeneous
behavior of the myocard into account. Three micro-structural,
orthogonal axes are distinguished: the fiber, sheet and sheet
normal axis. For each axis i
a set of parameters, ki, ai,
and βi,
describes its contribution to the strain energy density, called
pole-zero law:
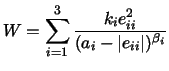 |
(15) |
with the diagonal components of the Green-Lagrange strain
tensor eii. The parameter ki
is set to zero, if eii
is negative. The strain energy density function W
is defined for |eii| < ai .
The function shows large values for eii
approaching ai ,
reflecting the steep rise in tension coming upon a strain
limit. The strain energy was extended by terms representing
the incompressibility of the myocard. The energy does not
comprise shear and viscoelastic effects. The parameterization
of the function W
was performed by uniaxial measurements of canine ventricle
in the different directions of the axes. Hereby, the parameters
of different regions in the myocard were collected.
3. Results
The developed numerical model has the purpose to achieve knowledge concerning
the cardiac deformation and its influence to the initiation and propagation
of electrical excitation and to the force development. The model combines
and extends the presented cellular and macroscopic models. It consists
of
-
a single cell electrophysiological model with stretch dependent behavior
-
an extended bidomain model taking stretch into account
-
a single cell model of the force development with inclusion of stretch
effects
-
an elastomechanical model
3.1 Modeling of the Cardiac Electromechanics
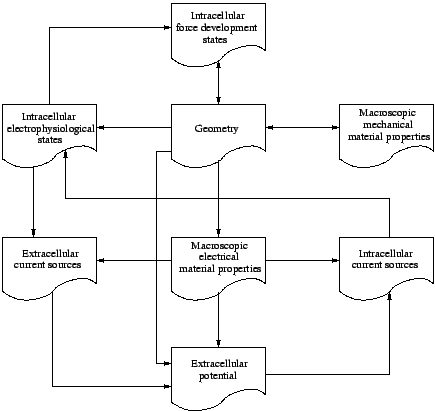
Figure 8: Modeling of cardiac electromechanics.
The interdependencies of the different data are depicted in figure 8.
As an electrophysiological model the modified Noble-Varghese-Kohl-Noble
model was used [Noble et al., 1998], whereby stretch
dependent ion channels were included [Sachse et al.,
2001]. The intercellular electrical coupling through the gap junctions
and extracellular space was performed with the extended bidomain model
[Sachse et al., 2000]. The engaged force model
was the Rice-Winslow-Hunter model (type 3). The elastomechanical behavior
was modeled by numerical methods of continuum mechanics [Bathe,
1996] using the strain energy function proposed by [Hunter
et al., 1997].
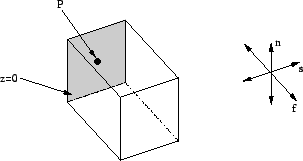
Figure 9: Model of myocardial area. The
electrical excitation is initiated by applying current at plane z=0. The
area is mechanically fixed at point P. The orientation of myocytes is indicated
by f, the sheet orientation by s, and the sheet normal by n.
3.2 Simulations
Simulations with the electromechanical model of a myocardial area were performed.
The results were visualized with surface based techniques
(figures 10 and 11).
The applied anatomical model consisted of 20 × 20 × 20 cubic
voxels, each with a size of 0.1 mm × 0.1 mm × 0.1 mm
(figure 9). The activation starts at time
0 ms by application of a sufficient electrical current at plane z = 0.
The principal axis of myocytes f was chosen parallel to the
z-axis. The lamination of the myocytes is determined by the
sheet orientation s and the sheet normal n. Transversal isotropy
of the electrical conductivities was set. Anisotropy of the
elastomechanical parameters and incompressibility was assumed.
The central position of the plane z=0 was fixed, i. e. the
displacements were set to zero. The electrophysiological modeling
(equation 1 and 2)
and the force development modeling (equation 11)
was performed with the Euler method using a time step of 20 μs
[ Press et al., 1992]. The bidomain
model used a Gauss-Seidel iteration every 20 μs
[ Press et al., 1992]. The deformation
was calculated with a time step of 1 ms.
The system of linear equations resulting from equation 13
was solved by the conjugate gradient method.
![\begin{figure}\center\subfigure[]{\epsfig{file=Vm.0.eps,width=.46\textwi......\subfigure[]{\epsfig{file=Vm.8.eps,width=.46\textwidth}}\end{figure}](08/img125.gif)
Figure 10: Transmembrane voltage at time
(a) 0 ms, (b) 3 ms, (c) 5 ms, and (d) 8 ms
in an anisotropic model of myocardial area. The model
consists of 20 × 20 × 20 cubic voxels with a size of
0.1 mm × 0.1 mm .
The simulations with the combined model show processes of different time
scale. The process of excitation propagation is rapidly spreading over
the myocardium (figure 10). The force development and
the resulting deformation follows with a significant delay (figure 11).
![\begin{figure}\center\subfigure[]{\epsfig{file=EM_Force0.eps,width=.49\subfigure[]{\epsfig{file=EM_Force5.eps,width=.49\textwidth}}\end{figure}](08/img126.gif)
Figure 11: Color-coded
normalized force and deformation at time
(a) 0 ms , (b) 50 ms, (c) 100 ms, (d) 150 ms, (e) 200 ms, and (f) 250 ms
in an anisotropic model of myocardial area. The model
consists of 20 × 20 × 20 cubic voxels with a size of
0.1 mm × 0.1 mm × 0.1 mm.
The central position of the plane z=0 was fixed, i. e.
the displacements were set to zero. The wire frame shows
the reference configuration.
4. Discussion and Conclusions
The presented model describes aspects of the electromechanical behavior
of a myocardial region. The model combines an electrophysiological, an
excitation propagation, a force development and an elastomechanical model.
The performed simulations illustrate effects of myocardial electromechanical
behavior. These effects are of great significance for the development of
realistic models of the whole heart. The presented combination of electrophysiological
and mechanical modeling allows the simulation of therapeutical interactions
with pharmaceutical, electrical and mechanical methods. This functionality
is of importance e.g. for studying of cardiac arrhythmias and for the computer
aided planning of surgical interventions. Further work will be focussed
on the inclusion of inertia and surface forces by blood pressure as well
as modeling of viscoelastic tissue properties. Of interest is also the
integration of models of the metabolism.
Acknowledgments
The authors want to thank Dr. P. Kohl, University Laboratory of Physiology,
Oxford, UK, for his help to parameterize the electrophysiologic model.
R. Mayer, Rechenzentrum, Univerität Karlsruhe (TH), supported our
work by providing the necessary visualization and computing resources.
References
Abrahams, C., Janicki, J. S., and Weber, K.
T.. Myocardial hypertrophy in macaca fascicularis: Structural remodeling
of the collagen matrix. Laboratory Investigation, 56:676-683. 1987
Bathe, K.-J. Finite Element Procedures in
Engineering Analysis. Prentice Hall, Englewood Cliffs/NJ. 1982.
Bathe, K.-J.. Finite Element Procedures.
Prentice Hall, Upper Saddle River, New Jersey. 1996
Beeler, G. W. and Reuter, H.. Reconstruction
of the action potential of ventricular myocardial fibres.J. Physiol.,
268:177-210. 1977
Bers, D. M. . Excitation-Contraction Coupling
and Cardiac Contractile Force. Kluwer Academic Publishers, Dordrecht,
Netherlands. 1991
Caulfield, J. B. and Borg, T. K. . The collagen
network of the heart. Laboratory Investigation, 403:364-372. 1979
Courtemanche, M., Ramirez, R. J., and
Nattel, S. . Ionic mechanisms underlying human atrial action potential
properties: insights from a mathematical model. Am. J. Physiol.,
27544:H301-H321. 1998
Dellmar, M., Morley, G. E., Ek-Vitorin, J. F.,
Francis, D., Homma, N., Stergiopoulos, K., Lau, A., and Taffet, S. M..
Intracellular regulation of the cardiac gap junction channel connexin43.
In Zipes, D. P. and Jalife, J., editors, Cardiac Electrophysiology.
From Cell to Bedside, chapter 15, pages 26-132. W. B. Saunders Company,
Philadelphia. 1999
Demir, S. S., Clark, J. W., Murphey, C. R., and
Giles, W. R.. A mathematical model of a rabbit sinoatrial node cell. Am.
J. Physiol., 35:832-852. 1994
Demir, S. S., O'Rourke, B., Tomaselli, G. F.,
Marbán, E., and Winslow, R. L.. Action potential variation in canine
ventricle: A modeling study. In Proc. Computers in Cardiology, volume
23, pages 221-224. 1996
Dhein, S.. Cardiac Gap Junctions. Karger.
1998
DiFrancesco, D. and Noble, D. A model of
cardiac electrical activity incorporating ionic pumps and concentration
changes. Phil. Trans. R. Soc. Lond., 307:353-398. 1985
Earm, Y. E. and Noble, D. A model of single atrial
cell: Relation between calcium current and calcium release. Proc. R.
Soc. Lond., 240:83-96. 1990
Eifler, W. J. and Plonsey, R.. A cellular model
for the simulation of activation in the ventricular myocardium. J. Electrocardiology,
82:117-128. 1975
FitzHugh, R. A. Impulses and physiological
states in theoretical models of nerve membran. Biophys J, 1:445-466.
Forbes, M. S. and Sperelakis, N. Intercalated
discs of mammalian heart: A review of structure and function. Tissue
and Cell, 175:605-648. 1985.
Henriquez, C. S., Muzikant, A. L., and Smoak,
C. K. Anisotropy, fiber curvature and bath loading effects on activation
in thin and thick cardiac tissue preparations: Simulations in a three-dimensional
bidomain model. J. Cardiovascular Electrophysiology, 75:424-444.
1996.
Henriquez, C. S. and Plonsey, R. A bidomain
model for simulating propagation in multicellular cardiac tissue. In Proc.
of the Annual International Conference of the IEEE Engineering in Medicine
and Biology Society, volume 4, page 1266. 1989.
Hilgemann, D. W. and Noble, D. Excitation-contraction
coupling and extracellular calcium transients in rabbit atrium: Reconstruction
of basic cellular mechanisms. Proc. R. Soc. Lond., 230:163-205.
1987.
Hodgkin, A. L. and Huxley, A. F. A quantitative
description of membrane current and its application to conduction and excitation
in nerve. J. Physiol, 177:500-544. 1952.
Hooke, N., Henriquez, C. S., Lanzkron, P., and
Rose, D. Linear algebraic transformations of the bidomain equations: Implications
to numerical methods. Crit Rev Biomed Eng, 120:127-145. 1992.
Hoyt, R. H., Cohen, M. L., and Saffitz, J. E.
Distribution and three-dimensional structure of intercellular junctions
in canine myocardium. Circ Res., 64:563-574. 1989.
Hunter, P., Nash, M. P., and Sands, G. P.Computational
electromechanics of the heart. In Panfilov, A. V. and Holden, A. V., editors,
Computational
Biology of the Heart, pages 345-408. John Wiley & Sons, Chichester.
1997.
Hunter, P. J., McCulloch, A. D., and ter Keurs,
H. E. D. J. Modelling the mechanical properties of cardiac muscle. Prog.
Biophys. Mol. Biol., 00:1-44. 1998.
Jafri, M. S., Rice, J. J., and Winslow,
R. L. Cardiac Ca2+
dynamics: The roles of ryanodine receptor adapation and
sarcoplasmic reticulum load. Biophysical J, 74:1149-1168.
1998.
Jongsma, H. J. and Rook, M. B. Biophysics of
cardiac gap junction channels. In Zipes, D. P. and Jalife, J., editors,
Cardiac
Electrophysiology. From Cell to Bedside, chapter 14, pages 119-125.
W. B. Saunders Company, Philadelphia. 1999.
Ju, H. and Dixon, I. M. C. Extracellular matrix
and cardiovascular diseases. Can. J. Cardiol., 1212:1259-1267. 1996.
Killmann, R., Wach, P., and Dienstl, F. Three-dimensional
computer model of the entire human heart for simulation of reentry and
tachycardia: Gap phenomenon and Wolff-Parksinson-White syndrome. Basic
Research in Cardiology, 865. 1991.
Landesberg, A. and Sideman, S. Coupling
calcium binding to troponin C and cross-bridge cycling in skinned cardiac
cells. Am. J. Physiol., 266:H1260-H1271. 1994a.
Landesberg, A. and Sideman, S. Mechanical
regulation of cardiac muscle by coupling calcium kinetics with cross-bridge
cycling: A dynamic model. Am. J. Physiol., 267:H779-H795. 1994b.
LeGrice, I. J., Smaill, B. H., Chai, L. Z.,
Edgar, S. G., Gavin, J. B., and Hunter, P. J. Laminar structure of the
heart: Ventricular myocyte arrangement and connective tissue architecture
in the dog. Am. J. Physiol., 269:H571-H582. 1995.
Luo, C.-H. and Rudy, Y. A model of the ventricular
cardiac action potential. Circ. Res., 686:1501-1526. 1991.
Luo, C.-H. and Rudy, Y. A dynamic model of the
ventricular cardiac action potential: I. simulations of ionic currents
and concentration changes. Circ. Res., 746:1071-1096. 1994a.
Luo, C.-H. and Rudy, Y. A dynamic model of the
ventricular cardiac action potential: II. afterdepolarizations, triggered
activity, and potentiation. Circ. Res., 746:1097-1113. 1994b.
McAllister, R. E., Noble, D., and Tsien,
R. W. Reconstruction of the electrical activitity of cardiac purkinje fibres.
J.
Physiol., 251:1-59. 1975.
Noble, D. A modification of the hodgkin-huxley
equation applicable to Purkinje fibre action and pacemaker potentials.
J.
Physiol., 160:317-352. 1962.
Noble, D., Varghese, A., Kohl, P.,
and Noble, P. Improved guinea-pig ventricular cell model
incorporating a diadic space, IKr
and IKs,
and length- and tension-dependend processes. Can. J.
Cardiol., 141:123-134. 1998.
Nygren, A., Fiset, C., Firek, L., Clark, J.
W., Lindblad, D. S., Clark, R. B., and Giles, W. R. Mathematical model
of an adult human atrial cell. Circ. Res., 82:63-81. 1998.
O'Rourke, B., Kass, D. A., Tomaselli, G. F.,
Kaab, S., Tunin, R., and Marbán, E. Mechanisms of altered excitation-contraction
coupling in canine tachycardia-induced heart failure, I experimental studies.
Circ.
Res, 845:562-570. 1999.
Panfilov, A. V. Three-dimensional wave propagation
in mathematical models of ventricular fibrillation. In Zipes, D. P. and
Jalife, J., editors, Cardiac Electrophysiology. From Cell to Bedside,
chapter 31, pages 271-277. W. B. Saunders Company, Philadelphia. 1999.
Press, W. H., Teukolsky, S. A., Vetterling, W.
T., and Flannery, B. P. Numerical Recipes in C. Cambridge University
Press, Cambridge, New York, Melbourne, 2 edition. 1992.
Priebe, L. and Beuckelmann, D. J. Simulation
study of cellular electric properties in heart failure. Circ. Res.,
82:1206-1223. 1998.
Rice, J. J., Jafri, M. S., and Winslow, R. L.
Modeling short-term interval-force relations in cardiac muscle. Am.
J. Physiol. Circ. Heart., 278:H913-H931. 2000.
Rice, J. J., Winslow, R. L., and Hunter, W. C.
Comparison of putative cooperative mechanisms in cardiac muscle: length
dependence and dynamic responses. Am. J. Physiol. Circ. Heart.,
276:H1734-H1754. 1999.
Rogers, J. M. and McCulloch, A. D. A collocation-Galerkin
finite element model of cardiac action potential propagation. IEEE Transactions
on Biomedical Engineering, 418:743-757. 1994.
Rudy, Y. and Quan, W. Mathematical model of reentry
of cardiac excitation. In Proc. Computers in Cardiology, volume
16, pages 135-136. 1989.
Sachse, F. B. Modeling of the mammalian heart.
Universität Karlsruhe TH, Institut für Biomedizinische Technik.
Habilationsschrift, in press. 2001.
Sachse, F. B., Riedel, C., Werner, C. D., and
Seemann, G. Stretch activated ion channels in myocytes: parameter estimation,
simulations and phenomena. In Proc. 23th Conf. IEEE Eng. in Med. and
Biol. in press. 2001.
Sachse, F. B., Seemann, G., Riedel, C., Werner,
C. D., and Dössel, O. Modeling of the cardiac mechano-electrical feedback.
In CardioModel 2000, Computer Models of the Heart: Theory and Clinical
Application, volume 2-2. International Journal of Bioelectromagnetism.
ISSN 1456-7865. 2000.
Saffitz, J. E. and Yamada, K. A. Gap junction
distribution in the heart. In Zipes, D. P. and Jalife, J., editors, Cardiac
Electrophysiology. From Cell to Bedside, chapter 21, pages 271-277.
W. B. Saunders Company, Philadelphia. 1999.
Saxberg, B. E. H. and Cohen, R. J. Cellular
automata models of cardiac conduction. In Glass, L., Hunter, P., and McCulloch,
A., editors, Theory of Heart, pages 437-476. Springer, Berlin, Heidelberg,
New York. 1991.
Sepulveda, N. G. and Wikswo, J. P. Bipolar
stimulation of cardiac tissue using an anisotropic bidomain model. J.
Cardiovasc. Electrophysiol., 53:258-267. 1994.
Siregar, P., Sinteff, J. P., Chahine, M., and
Beux, P. L. A cellular automata model of the heart and its coupling with
a qualitative model. Computers and Biomedical Research, 29:222-246.
1996.
Siregar, P., Sinteff, J. P., Julen, N., and
Beux, P. L. An interactive 3D anisotropic cellular automata model of the
heart. Computers and Biomedical Research, 31:323-347. 1998.
Spach, M. S. and Heidlage, J. F. The stochastic
nature of cardiac propagation at a microscopic level. Circ. Res.,
763:118-130. 1995.
Spach, M. S., Heidlage, J. F., and Dolber, P.
C. The dual nature of anisotropic discontinous conduction in the heart.
In Zipes, D. P. and Jalife, J., editors, Cardiac Electrophysiology.
From Cell to Bedside, chapter 25, pages 213-222. W. B. Saunders Company,
Philadelphia. 1999.
Streeter, D. D. . Gross morphology and fiber
geometry of the heart. In Bethesda, B., editor, Handbook of Physiology:
The Cardiovascular System, volume I, pages 61-112. American Physiology
Society. 1979
Streeter, jr., D. D. and Bassett, D. L. An
engineering analysis of myocardial fiber orientation in pig's left ventricle
in systole. Anatomical Record, 155:503-512. 1966.
Virag, N., Blanc, O., Vesin, J. M., Koerfer,
J., and Kappenberger, L. Study of the mechanisms of arrhythmias in an anatomical
computer model of human atria. In Proc. Computers in Cardiology,
volume 26, pages 113-116. 1999.
Weber, K. T., Sun, Y., Tyagi, S. C., and Cleutjens,
J. P. M. Collagen network of the myocardium: Function, structural remodeling
and regulatory mechanisms. J. Mol. Cell. Cardiol., 26:279-292. 1994.
Wei, D., Okazaki, O., Harumi, K., Harasawa, E.,
and Hosaka, H. Comparative simulation of excitation and body surface electrocardiogram
with isotropic and anisotropic computer heart models. IEEE Transactions
on Biomedical Engineering, 424:343-357. 1995.
Werner, C. D., Sachse, F. B., and Dössel,
O. Applications of the visible man dataset in electrocardiology: Simulation
of the electrical excitation propagation. In Proc. Second Users Conference
of the National Library of Medicine's Visible Human Project, pages
69-79. 1998.
White, E., Guennec, J.-Y. L., Nigretto, J. M.,
Gannier, F., Argibay, J. A., and Garnier, D. The effects of increasing
cell length on auxotonic contractions; membrane potential and intracellular
calcium transients in single guinea-pig ventricular myocytes. Experimental
Physiol., pages 65-78. 1993.
Winslow, R. L., Rice, J. J., Jafri, S., Marbán,
E., and O'Rourke, B. Mechanisms of altered excitation-contraction coupling
in canine tachycardia-induced heart failure, II model studies. Circ.
Res., 84:571-586. 1999.
Yeager, M. Molecular biology and structure of
cardiac gap junction intercellular channels. In Zipes, D. P. and Jalife,
J., editors, Cardiac Electrophysiology. From Cell to Bedside, chapter
4, pages 31-40. W. B. Saunders Company, Philadelphia. 1999.
|

 Home
Current Issue
Table of Contents
Home
Current Issue
Table of Contents

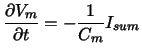
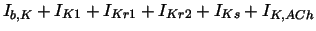
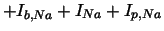
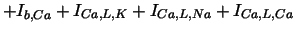
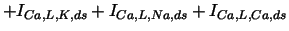
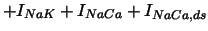
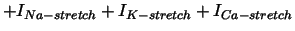
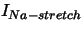 ,
,
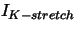 ,
,
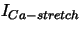 ,
,





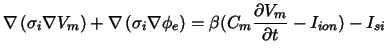

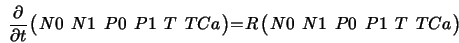
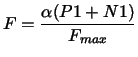
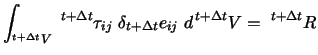
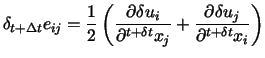
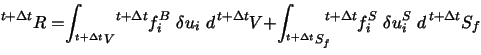
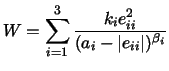


![\begin{figure}\center\subfigure[]{\epsfig{file=Vm.0.eps,width=.46\textwi......\subfigure[]{\epsfig{file=Vm.8.eps,width=.46\textwidth}}\end{figure}](08/img125.gif)
![\begin{figure}\center\subfigure[]{\epsfig{file=EM_Force0.eps,width=.49\subfigure[]{\epsfig{file=EM_Force5.eps,width=.49\textwidth}}\end{figure}](08/img126.gif)