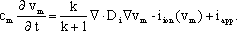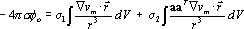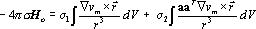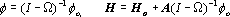Correspondence: Kim Simelius,
Nokia Mobile Phones, P.O. Box 68, FIN-33721 Tampere, Finland.
E-mail: kim.simelius@nokia.com,
phone & SMS: +358 50 303 8377, fax: +358 7180 46847,
mobile fax: +358 50 8303 8377
1. Introduction
While there is hardly any lack of electrocardiographic
or magnetocardiographic data, detailed data on the anatomy
and physiology of the conduction system or cellular interactions
in the myocardium are scarce [Durrer et al., 1979]. We may
even have access to high-quality recorded data on the endocardial
and epicardial potentials during cardiac activation [Gepstein
et al., 1997], but we are left to guess how exactly the
propagation through the myocardium takes place. However,
a ventricular model that produces a correct normal activation
sequence is a prerequisite for simulating pathological conditions,
such as ischemia, infarction or ventricular arrhythmias
[Berenfeld and Jalife, 1998]. Therefore, a comprehensive
model should feature an anatomically accurate geometry,
intramural fibrous structure, and a conduction system, and
the action potential of a single cell should be modeled
realistically. As if there were not enough difficulty in
obtaining accurate data to base the modeling on, implementing
the models themselves is a challenge.
First idealized models describing the normal activation
sequence in the human heart were reported over two decades
ago [Ritsema van Eck, 1972; Miller and Geselowitz, 1978].
Mainly due to computational limitations, the models did
not include myocardial anisotropy or physiological propagation.
On the basis of anisotropic bidomain theory [Colli Franzone
et al., 1983] and cellular automata theory [Toffoli et al.,
1987], development of more realistic whole-heart models
has become feasible [Leon and Horáček, 1991]. In this
paper, we concentrate on the modeling of the interacting
cellular elements and the bidomain equations, implementation
of the conduction system and the computation of extracardiac
electromagnetic fields. As our model of the action potential
is still primitive, we are also limiting ourselves here
to discussing the ventricular depolarization.
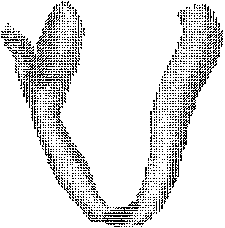
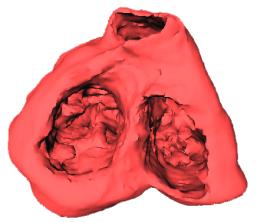
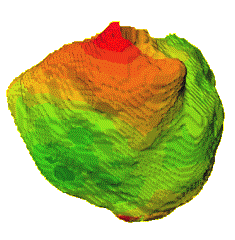
Figure 1. Left: Fiber
arrangement on an yz-cross section approximately in the
middle of the ventricles. The principal fiber directions
are marked with arrows. The angles, measured from the local
tangent vector at each element, rotate from about -50 degrees
on the endocardium to about +45 degrees on the epicardium.
Right: Three-dimensional presentation of the ventricles
as seen from the base of the heart.
2. Methods
2.1. Anisotropic Propagation Model and the Cellular Elements
The propagation model consists of 2.000.000 excitable elements
comprising the conduction system and the myocardium and
8.000.000 non-excitable elements making up the intra- and
extracardiac volumes. The elements are located on a cubic
lattice with 0.5-mm spacing [Hren et al., 1998]. The geometry
was reconstructed from 1-mm sections of the human heart
[Ritsema van Eck, 1972]. The assignment of the principal
fiber direction was performed separately for left-ventricular,
right-ventricular and papillary-muscle cells, with the fiber
direction rotating from endocardium to epicardium [Streeter,
1979]. Fig. 1 illustrates the amount of anatomical detail
that was included in the model.
Our hybrid model of the anisotropic myocardium describes
the subthreshold behaviour of the elements according to
the bidomain theory [Colli Franzone et al., 1983],
while in the suprathreshold region the elements behave as
cellular automata [Toffoli and Margolus, 1987]. Each element
is assigned a specific type and a vector of local fiber
direction based on anatomical data [Leon and Horacek, 1991;
Taccardi et al., 1994]. Each element can be in one of four
macrostates, corresponding to different phases of an action
potential: resting, excitatory, absolute refractory, or
relative refractory. During simulation, elements undergo
a series of macrostate transitions, which have been described
in detail in [Nenonen et al., 1991]. The subthreshold transmembrane
potential, vm is calculated from the generalized
cable equation, derived from the anisotropic bidomain theory
[Colli Franzone et al., 1983]:
| |
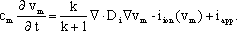 |
(1) |
Here we assume that the assumption of equal anisotropy
ratios is valid [Leon and Horacek, 1991; Nenonen et al.,
1991]. In Eq. 1, cm is the specific membrane
capacitance, Di is the intracellular conductivity
tensor, and iion and iapp are ionic
and applied currents, respectively. Above a threshold value,
the cell enters the absolute refractory state and vm
is estimated from a predefined function consisting of an
upshoot part and a logarithmic plateau phase [Nenonen et
al., 1991]. Physiological parameters like the conductivities
and the specific membrane capacitance for the model are
adopted from literature [Colli Franzone et al., 1983; Roth,
1991].
The correct discretization of Eq. 1 is crucial for a directionally
unbiased activation. Since the cell size is reasonably large
compared to the dimensions of the ventricular structures,
the estimation of the divergence of the current density
is done in a 3x3 cube of cells. Now, using traditional differentiation
stencils that misemphasize the diagonal directions can,
e.g., turn the activation in the direction of the coordinate
axes. Here, a method similar to the “natural neighbors”
method [Braun and Sambridge, 1995] can be used to alleviate
the problem.
2.2. Extracardiac Field Calculation
In the forward computations, the ventricles are assumed
to be embedded in a torso-shaped piecewise homogeneous volume
conductor, including the lungs and the intracardiac blood
masses. The infinite medium potential fo in the
extracardiac region is determined from the discretized equation
| |
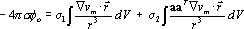 |
(2) |
where the integrals are evaluated over the ventricular
volume, vm is as in Eq. 1, a is the local
direction of the fiber axis, s is the extracellular conductivity
(s = 2.0 mS/cm), s1 is the transverse conductivity
(s1 = 0.5 mS/cm) and s2 is the difference
between the axial and transverse conductivity (s2
= 1.5 mS/cm) [Nenonen et al., 1991]. The first term of the
potential in Eq. 2 represents the contribution of the isotropic
component and the second term accounts for the anisotropic
properties of the cardiac tissue. The extracardiac magnetic
field Ho is evaluated from a corresponding
equation,
| |
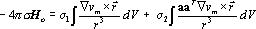 |
(3) |
To compute the body surface potential and magnetic field
in the inhomogeneous torso, a "fast forward solution"
is used [Nenonen et al., 1991]:
| |
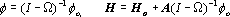 |
(4) |
In Eq. 4, W is a geometry matrix consisting of the solid
angles subtended by a surface element at each node, and
A is another (vector-value) geometry matrix taking
into account the influence of the conductivity change surfaces
on the magnetic field (for details, see [Nenonen et al.,
1991b]). The anisotropy of the volume conductor can be estimated
by the thorax extension method [Lorange and Gulrajani, 1993].
When the Eqs. 2 and 3 are discretized and subsequently
the integration (or summation) is carried out, special care
has to be taken that the elementary current sources do not
contribute to the fields multiple times. If one aims at
a good approximation of the elementary sources, a differentiation
stencil of 2x2 or 3x3 elements needs to be used. However,
since the integrand is vector-valued, any contribution from
the diagonal element pairs easily misweights the elementary
dipoles. Some error is also always present due to the fact
that the fiber direction used for estimating the (finite-size)
dipole at a certain location is computed from the starting
point of the dipole, not the center.
2.3. Conduction System
The organization of the conduction system was created by
using information available from textbooks on human anatomy
and other literature on the topic [Berenfeld and Jalife,
1998; Demoulin and Kulbertus, 1982; Durrer et al., 1979;
Tawara, 1906]. In anatomical studies, the left bundle branch
has been found to be a sheet-like structure that covers
a large portion of the interventricular septum. The fascicles
in the branch are highly interconnected. The anterior rim
of the left bundle travels towards the left anterior papillary
muscle, while the posterior rim is oriented toward the left
posterior papillary muscle. The right bundle branch usually
begins from the most distal part of the His bundle. It courses
subendocardially and intramyocardially towards the right
anterior papillary muscle. At the papillary muscle, it divides
into fascicles that continue as the right Purkinje network
leading the activation to the posterior papillary muscle
and the right ventricular free wall. The His bundle continues
on both sides as the Purkinje network that contains the
Purkinje-myocardial junction (PMJ) sites. On the right,
the most prominent feature of the conduction system is the
single bundle that carries the activation from the septum
to the free wall and the papillary muscles along the moderator
band. On the left, there are three major areas of activation:
the septum and the inferior and superior free wall.
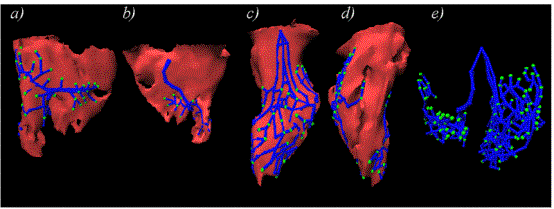
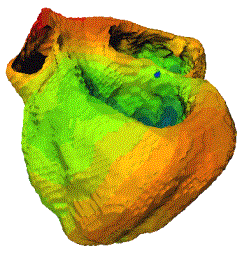
Figure 2. The geometry
of the conduction system observed from different angles.
The right branch of the His bundle (not shown) connects
the topmost nodes of the left and right conduction system.
In the right ventricle, the moderator band contains the
conduction system activating the RV free wall and the papillary
muscles. The RV free wall (a) and septal (b) views show
the right conduction system, consisting of one main bundle
with several smaller branches.The views of LV septum (c)
and LV free wall (d) show the more complicated structure
of the left conduction system. Notice the fan-like structure
of the septal conduction system in (c), and the vertical
spiral gap in the conduction system in (d). The PMJs are
shown as light green spheres.
The gross geometry of the conduction system is shown in
Fig. 2. To obtain correct electromagnetic fields on the
body surface, the conduction system has to be carefully
tuned. The right balance between the activation times in
the left and the right ventricle is necessary to create
a correct 12-lead ECG and VCG. On the other hand, to obtain
a ventricular activation sequence corresponding to measured
data [Durrer et al., 1979], the correct location and number
of the Purkinje-myocardial junction sites is crucial. In
the course of tuning, it often happens that these two requirements
cannot be fulfilled by the same conduction system model.
Therefore, the tuning of the model was performed sequentially
for the isochrones of Durrer, the body surface potential
and magnetocardiographic maps and finally for the 12-lead
ECG and the vectorcardiogram.
3. Results
3.1. Propagation in the 3D Ventricles - Isochrones
The simulated activation sequence resulting from the activation
of the conduction system is shown in Fig. 3. The ventricular
activation starts in the left ventricular septum (layer
110), matched by a right ventricular septal activation 20
ms later (layers 70-90). Almost simultaneously with the
RV septal activation, the inferior (in body coordinates)
and anterior LV activation appear (layers 90-110). These
are followed by the activation of the RV free wall (layers
90-130). The RV and LV breakthroughs take place at 30 ms
and 45 ms, respectively. In the final stages of the QRS,
activation propagates through the posterior LV free wall
and the pulmonary conus. The result agrees well with isochrones
obtained from an isolated human heart [Durrer et al., 1979].
Some minor differences can be seen in the activation of
the right ventricle. However, based on high interindividual
variability in Durrer's work and other literature sources
describing the human ventricular activation [van Dam, 1976],
the RV activation sequence seems to be somewhat unclear.
Therefore, we trusted our model of the conduction system
to be correct, since the electromagnetic fields produced
by the activation were in agreement with our recordings.
A three-dimensional view of the activation isochrones is
shown in Fig. 4.

Figure 3. Simulated
activation isochrones. The 20 layers are with a spacing of
10 mm, and correspond to those presented
by Durrer et al. The isochrone spacing is 5 ms.
Figure 4. Three-dimensional
views of the simulated isochrones. a) Anterolateral apical
view of the right ventricle, b) oblique cross section across
the ventricles c) posterolateral basal view of the left
ventricle, d) a cross section showing the intramural activation
isochrones in both ventricles.
3.2. Simulated BSPM and MCG Patterns
BSPMs were evaluated on the nodes of a triangulated torso
model of an adult male, while the normal component of the
cardiomagnetic field (MCG) was computed on a plane array
in front of the chest. Fig. 4 shows simulated BSPM and MCG
distributions during the QRS complex. In BSPM, the initial
maximum resulting from left septal activation is anterior.
Then the minimum on the back moves upward and travels over
the right shoulder onto the right anterosuperior region,
indicating apical activation and masking of the left septal
activation by the corresponding right-septal activation.
The area of positive potentials then drifts to the back.
In the end, the activation propagates toward the pulmonary
conus, reflected as a superior positive potential in BSPM.
The sequence of MCG patterns essentially depicts the direction
of the frontal ECG vector, i.e. the direction of activation
in the plane of the frontal chest.
The mean body surface potential map series of healthy patients
displays a similar time development as the simulated BSPM.
What is noteworthy here again is the large interindividual
variability, which is certainly understandable. The diversity
of conduction systems is added to by the differences in
body shape, the volume conductor properties and even the
physiological state of the patient during the recording.
For example, the initial maximum may well be located on
the lower half of the chest, and the rotational movement
may take place from right to left on the frontal chest,
not diagonally. Against this, the simulated BSPM patterns
seem acceptable.
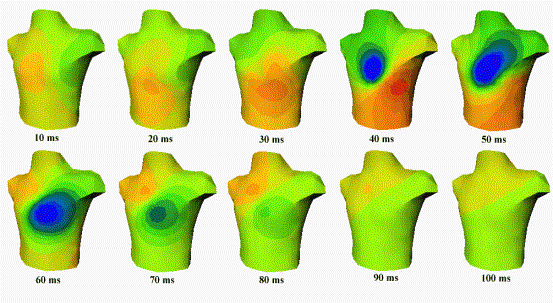
Figure 5. Simulated
BSPM distributions at 10 ms steps during the QRS complex.
The read and blue areas denote, respectively, positive and
negative values. At 50 ms, the positive area has moved to
the back (not shown on the series).
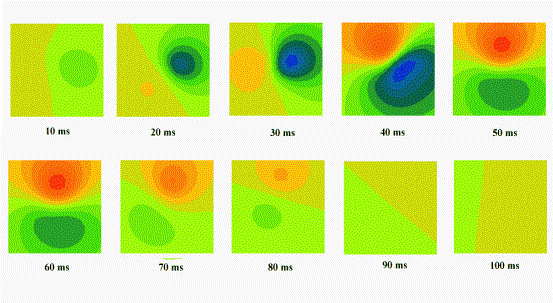
Figure 6. Simulated
MCG distributions at 10 ms steps during the QRS complex.
The red areas denote positive values, where the magnetic
flux enters the chest, while in blue areas the flux comes
out of the chest.
3.3. Simulated Electrocardiograms and Vectorcardiograms
The “normal” 12-lead electrocardiogram is even
more difficult to define than the normal BSPM, but some
common features should anyway be present in the ECG to fulfill
“normality”. The dominant features affecting all
leads are – as can be seen from the BSPMs – that,
in the beginning of the QRS complex, the left leg is positive
and in the end, the activation propagates towards the back.
This means that V1 and V2 should display a mainly negative
morphology (activation away from the electrode) preceded
by a small positive deflection. On the other hand, in the
leads V4, V5, V6, I, II and aVF, positivity is the dominating
feature. As can be seen, the simulated ECG fulfills these
criteria reasonably well. However, the detailed morphology
of the chest leads is somewhat incorrect, probably due to
thorax modeling problems.
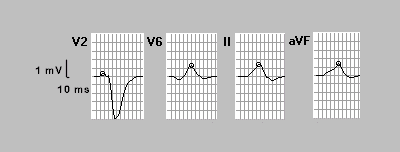
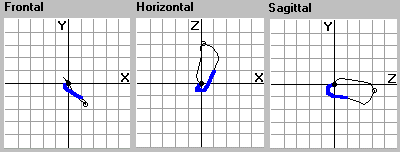
Figure 7. Simulated
ECG leads and the vectorcardiographic loops. Top: ECG leads
V2, V6, II and aVF. Bottom: the frontal, horizontal and
sagittal VCG loops. The potential grid spacing is 0.5 mV
and the time grid spacing (ECG) is 10 ms. Note how the frontal
VCG loop is directed towards the left leg.
The normal vectorcardiogram (VCG) is characterized by a
loop, whose average direction is to the left, down and slightly
back. The frontal loop is very thin, and the sagittal and
horizontal loops are somewhat wider. The sagittal and horizontal
loops are clearly counter-clockwise. The frontal loop defines
a clear “angle” that should be from –20o
to +110o from the vertical axis pointing down.
The simulated vectorcardiogram also follow this pattern
well.
4. Discussion
Information from anatomical and electrophysiological studies
on the conduction system are to some extent contradictory.
The modelling is further complicated by the fact that anatomical
information on the conduction system is old, and no digital
data are available. Furthermore, few thorough works on activation
sequences of the human heart exist.
Several details of the model algorithm are still under
discussion in the scientific community. It is still somewhat
unclear, whether simplifying the modelling of anisotropic
properties by the use of equal anisotropy ratios affects
the propagation significantly. The effect of thoracic and
cardiac muscle anisotropies on the forward solution has
also been discussed. However, more fundamental features
like transmurally varying cell properties may mask the effects
of anisotropy.
The activation sequence produced by our simulations was
compared to the activation sequence measured from the isolated
human heart. The simulated activation of the left ventricle
matched very well the recorded one, when the cross sections
are aligned. However, the data from Durrer et al.
almost completely lacks right septal activation, which is
necessary to produce correct ECG.
The simulated body surface potential pattern corresponds
to our recorded BSPMs with the exception of two features:
the initial positive area on the anterior chest moves slightly
too quickly to the back and the late activation (positive)
of the pulmonary conus is too strong on the anterior chest.
Both these features may be attributed to inaccurate positioning
of the ventricular model within the thorax. The magnetocardiographic
maps are also consistent with recorded data. The general
outline on the simulated 12-lead ECG is normal, and the
vectorcardiogram is completely in the normal limits, the
frontal angle being close to 60o.
Acknowledgements
This research was partly financed by the Jenny and Antti
Wihuri Foundation and the Emil Aaltonen Foundation. The
first author also wishes to express his gratitude to Nokia
Mobile Phones for supporting the work.
References
Berenfeld O and Jalife J. Purkinje-muscle reentry as a
mechanism of polymorphic ventricular arrhythmias in a 3-dimensional
model of the ventricles. Circ Res, 82:1063-1077,
1998.
Braun J and Sambridge M. A numerical method for solving
partial differential equations on highly irregular evolving
grids. Nature, 376:655-660, 1995.
Colli Franzone P, Guerri L and Viganotti C. Oblique dipole
layer potentials applied to electrocardiology. J Math
Biol, 17:93-124, 1983.
Colli Franzone P, Guerri L and Taccardi B. Spread of excitation
in a myocardial volume: Simulation studies in a model of
anisotropic ventricular muscle activated by point stimulation.
J Cardiovasc Electrophysiol, 4:144-160, 1993.
van Dam RTh. Ventricular activation in human and canine
bundle branch block. The Conduction System of the Heart:
Structure, Function and Clinical Implications, 377--392,
Lea & Febiger, Philadelphia, 1976.
Demoulin JC and Kulbertus H. Pathological correlates of
intraventricular conduction disturbances. Cardiac Electrophysiology
Today. Academic Press, 1982.
Durrer D, van Dam RTh, Freud GE, Janse MJ, Meijler FL and
Arzbaecher RC. Total excitation of the isolated human heart.
Circulation, 41:899-912, 1979.
Gepstein L, Hayam G and Ben-Haim S. A novel method for
nonfluoroscopic catheter-based electroanatomical mapping
of the heart: In vitro and in vivo accuracy results. Circulation,
95:1611-1622, 1997.
Hren R, Nenonen J and Horáček BM. Simulated epicardial
potential maps during paced activation reflect myocardial
fibrous structure. Ann Biomed Eng, 26:1022-1035,
1998.
Leon LJ and Horacek BM. Computer model of excitation and
recovery in the anisotropic myocardium. J Electrocardiol,
24:1-41, 1991.
Lorange M and Gulrajani RM. A computer heart model incorporating
anisotropic propagation. I. Model construction and simulation
of normal activation. J Electrocardiol, 26:245-261,1993.
Miller WT III and Geselowitz DB. Simulation studies of
the electrocardiogram I: The normal heart. Circ Res,
43:301-315, 1978.
Nenonen J, Edens JA, Leon LJ and Horáček BM. Computer
model of propagated excitation in the anisotropic human
heart:
I. Implementation and algorithms. In Computers in Cardiology,
IEEE Computer Society Press, Los Alamitos, 1991, 545-548.
Nenonen J, Purcell CJ, Horáček BM, Stroink G and Katila
T. Magnetocardiographic functional localization using a
current dipole in a realistic torso. IEEE Trans Biomed
Eng, 38:658-664, 1991.
Ritsema van Eck HJ. Digital-computer simulation of cardiac
excitation and repolarization in man. PhD Thesis, Dalhousie
University, Halifax, Canada.
Roth BJ. Action potential propagation in a thick strand
of cardiac muscle. Circ Res, 68:162-173, 1991.
Streeter DD Jr. Gross morphology and fiber geometry of
the heart. Handbook of physiology - Section2: The cardiovascular
system. Volume 1: the heart, 61-112, Am Physiol Soc, Bethesada
MD, 1979.
Taccardi B, Macchi E, Lux RL, Ershler PE, Spaggiari S,
Baruffi S and Vyhmeister Y. Effect of myocardial fiber direction
on epicardial potentials. Circulation, 90:3076-3090,
1994.
Tawara S. Das Reizleitungssystem des Säugetierherzens.
Eine anatomisch-histologische Studie über das Atrioventrikularbündel
und die Purkinjeschen Fädern. Gustav Fischer Verlag, Jena,
1906.
Toffoli T and Margolus N. Cellular Automata Machines, A
New Environment For Modeling. MIT Press, Cambridge, MA,
1987.



 Home
Current Issue
Table of Contents
Home
Current Issue
Table of Contents