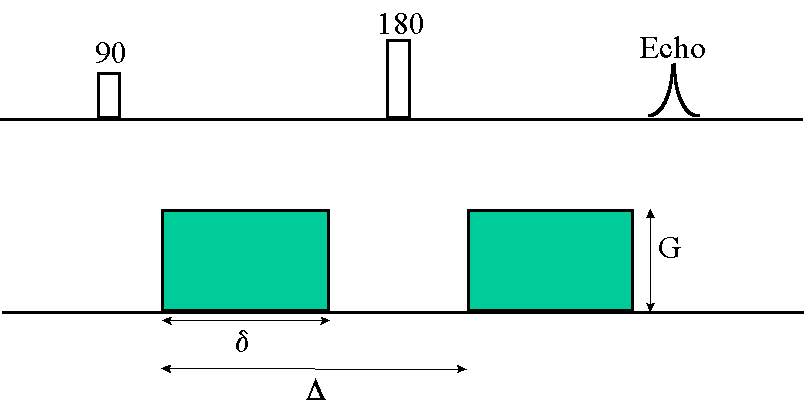H. J. Aronena,b, A. Korvenojaa, S. Martinkauppia, J. Perkiöa,
J. Karonenb,
and S. Carlsonc
1. Introduction
During the last ten years various functional magnetic resonance techniques have been developed which can be
used clinical patient studies. Two main prerequisites have enabled to bring these techniques into clinical studies.
First, the development of ultrafast imaging, especially echo planar imaging (EPI), nowadays available in 1.5 Tesla
scanners. Originally, the technique was proposed as early as 1977 by Mansfield . EPI is a method by which spatial
encoding for complete images is performed in a time shorter than, or comparable to, tissue T2 decay times. Because
it is fundamentally just a spatial-encoding technique, EPI has the advantage of a rather similar image contrast
than conventional MR imaging. The second prerequisite for the development of fMRI has been better understanding
of the basic relaxation phenomena, especially susceptibility contrast . In this review we discuss the basics and
clinical applications of three important functional magnetic resonance imaging techniques: diffusion and perfusion
imaging, and brain activation studies.
2. Diffusion imaging
The basic principles of measuring diffusion by nuclear magnetic resonance were laid out already in the 1950s
by Stejskal and Tanner . In MRI, these principles are utilized in studying the molecular diffusion of water - in
diffusion weighted imaging (DWI). A proton of a water molecule experiences slightly different field strength dependent
on its spatial location because of local inhomogeneities in magnetic field. Because the field strength is directly
proportional to the precession frequency, the moving spins acquire phase shift compared to static spins. Thus,
if a normal spin echo imaging sequence is used the echo is formed normally for static spins but only a weak signal
is detected for moving spins because they do not reach their initial phase at the time of echo. Although, a spin
echo sequence is sensitive to diffusion the effect is usually too small to be detected. However, diffusion sensitivity
can be increased by diffusion gradients put symmetrically around the 180 degree pulse (Figure 1). Diffusion sensitivity
is expressed in the so called b-value
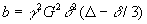
where G is the gradient amplitude, δ is the length of the gradient, Δ
is the temporal difference of the two gradient pulses, and γ is the gyromagnetic ratio
of a proton. The greater the b-value the more diffusion weighting there is. Since signal intensity is
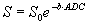
where S0 is the signal intensity without diffusion weighting and ADC the apparent diffusion coefficient,
the signal decreases with increasing diffusion weighting. The ADC-value can be calculated by using two or more
b-values, and thus diffusion can be measured quantitatively.
Diffusion of water in a tissue is restricted and anisotropic which makes it dependent on the direction of measurement.
Usually diffusion is measured in three orthogonal directions and the mean value used to approximate diffusion in
any direction. This mean is the trace of the diffusion tensor a mathematical object that fully describes the
dependence of diffusion on orientation. To determine the whole diffusion tensor a diffusion measurement is required
in at least six different directions. With a known diffusion tensor, diffusion anisotropy can quantified and there
are indications that this makes it possible to characterize e.g. the orientation of white matter tracts .
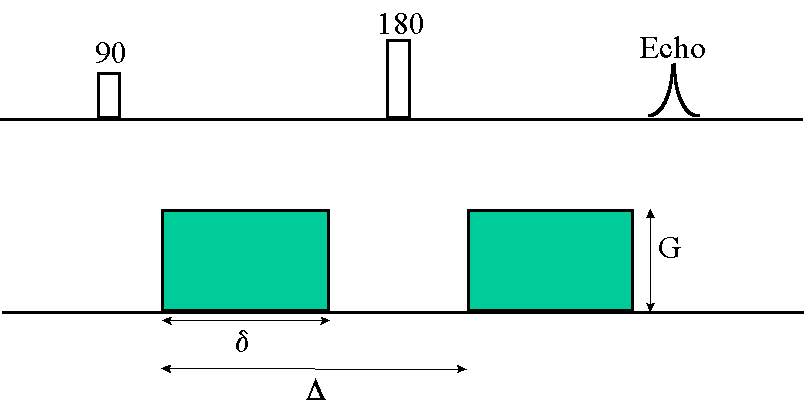
Figure 1. Diffusion weighted spin echo pulse sequence. Diffusion gradients (amplitude G, lenght
δ, interval Δ ) increase the
effect of magnetic field inhomogeneities and thus diffusion sensitivity.
2.1 Clinical applications
The ability of diffusion weighted imaging to detect changes in molecular diffusion of water can
be utilized in imaging of cerebral ischemia. When cerebral blood flow diminishes below a critical level, failure
of ionic pumps in the cell membrane to maintain ionic gradients leads to a net influx of water into the cell (cytotoxic
edema). In 1990, Moseley et al. reported that DWI was more sensitive than T2-weighted imaging in detecting early
cerebral ischemia after occlusion of the middle cerebral artery (MCA) in a cat stroke model . Reith et al. showed
DWI findings in rat 5 minutes after occlusion of the MCA . Hoehn-Berlage et al. showed that by measuring apparent
diffusion coefficient (ADC), one can detect a gradual increase of severity of ischemia from the periphery to the
core of the lesion . Determination of ADC provides a method to evaluate the severity of the ischemic lesion. Although
reperfusion has shown to cause recovery of ischemic lesions in animals , there has been few observations of recovery
in humans . Warach et al. studied 32 stroke patients with DWI and showed it to be more sensitive than T2-weighted
imaging also in humans . ADC can be helpful in determining the age of an ischemic lesion: ADC is decreased for
3-4 days from ictus after which a process called ´pseudonormalization´ occurs during days 5 to 10 .
In chronic infarct, the ADC is higher than in normal brain. ADC maps help to avoid wrong positive ischemic findings
a result of T2-shine-through, which is a problem in diffusion weighted raw images and trace images. Lutsep et al.
studied the clinical utility of DWI in assessment of ischemic stroke and find it superior to T2-weighted imaging
in acute situation Acute DWI lesion volumes have been shown to correlate with stroke scale scores and the final
infarct volume . Despite the observations that in experimental models of non-human stroke, DWI lesions can recover
with early reperfusion, lesions with decreased diffusion in human stroke are generally considered infarcted. DWI
is clinically applicable method that provides useful information about hyperacute ischemic stroke. In our material
in Kuopio University Hospital we have studied the natural course of ischemic lesion. We have observed that the
DWI lesion enlarges between the day of the ictus and the day after that, when the first DWI was performed approximately
10 hours from the onset of symptoms . A statistically significant enlargement is seen even between the lesion volume
on the second day and one week. This temporal course of the ischemic process supports the view that human ischemic
stroke with partial occlusion and possible clot lysis differs from experimental stroke with animal models.
3. Perfusion imaging
By combining the simultaneous use of paramagnetic contrast material and ultrafast imaging it is possible to
study the hemodynamics of normal and diseased human brain . Perfusion refers to blood circulation at the capillary
level. In perfusion weighted imaging (PWI) the first-pass of an intravascular, paramagnetic, non-diffusible contrast
agent through a region of interest, is studied. While in normal MR angiography the image voxel contains 100% of
flowing elements in PWI the percentage is only 2-4% which makes the technique very delicate. The magnetic susceptibility
difference between the capillaries filled with contrast agent and the surrounding tissue induces local magnetic
field gradients which alter the measured signal . In human brain the first-pass of the contrast agent bolus is
about 15 seconds which demands ultra-fast imaging in order to adequately sample the signal-time curve. MR signal
is converted into concentration-values by assuming a linear dependence between contrast agent concentration and
T2 relaxation rate. It is commonly believed that susceptibility contrast with a spin echo (SE) echo planar imaging
(EPI) sequence is specific to small vessels, in contrast to gradient echo EPI . Relative cerebral blood volume
(rCBV) can be determined by numerically integrating the concentration-time curve during first-pass of the bolus.
rCBV is a measure of the amount of capillaries in a volume element. Relative cerebral blood flow (rCBF) can be
determined if the arterial input function of the contrast agent is know . By deconvoluting the tissue concentration
time curve by the AIF, CBF and the residue function R(t) (which describes the fraction of injected tracer present
at the vasculature as a function of time) can be determined
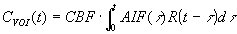 .
.
According to the central volume theorem, the mean transit time (MTT) of the contrast agent through
a voxel can be obtained as the relation of rCBV to rCBF. Recently, it has been proposed that in addition to relative
values, also absolute values can be obtained with PWI .
3.1 Clinical applications: Ischemic stroke
Imaging of brain perfusion by using single photon emission computed tomography (SPECT) and positron emission
tomography (PET) do not give morphological information about the brain, which seriously limits the clinical use
of these methods in daily practice. Perfusion weighted MR imaging provides an opportunity to image perfusion in
the same imaging session as DWI and conventional, more morphologically oriented MR imaging. Villringer et al. introduced
the susceptibility effect of gadolinium in 1988 . It is the basis of contrast agent bolus tracking technique, which
is at present clinically the most applicable PWI method. In the material of Warach et al. DWI and single slice
PWI were more accurate than conventional MRI in predicting clinical improvement . Studies combining DWI and PWI
have suggested that an acute infarct (detected by DWI) surrounded by hypoperfused zone (detected by PWI) could
especially be at risk of enlargement . In a study of ten patients by Tong et al. lesion sizes in DWI and PWI correlated
significantly with acute stroke scale score and final infarct size . They used time to peak maps and observed a
hypoperfusion area larger than or equal to the DWI changes in all patients. Barber et al. observed larger perfusion
defect in 11 out of 17 patients by using mean transit time maps and this perfusion-diffusion mismatch predicted
infarct expansion . Acute PWI and DWI lesion volumes correlated with clinical outcome. Lesion volumes detected
by PWI depend on the type of perfusion map used in volumetric analysis of hypoperfused tissue. In our series of
26 patients, the PWI lesion was larger than DWI lesion in 7 cases when measured from rCBV (relative cerebral blood
volume) maps, in 15 cases when measured from rCBF (relative cerebral blood flow) maps and in 18 cases when measured
from MTT (mean transit time) maps . In the study of Sorensen et al. rCBF maps combined with DWI predicted infarct
growth more often (12 of 15 subjects) than rCBV maps combined with DWI (8 of 15 subjects), whereas rCBV and DWI
lesion volumes showed better correlation with eventual infarct volume . Figure 2 shows a representative case with
acute perfusion-diffusion mismatch and subsequent infarct enlargement. The rCBF map shows a hypoperfusion area
that is of about the same size than the perfusion defect on SPECT. In our material, the perfusion-diffusion mismatch
volume on the first day predicted worsening in early clinical outcome assessed with National Institute of Health
Stroke Scale (NIHSS) . As a conclusion, combining perfusion and diffusion imaging seems to be a particularly effective
method to assess the early hemodynamic changes in the acute ischemic stroke.
Ultra fast MR imaging sequences have made it possible to combine DWI and PWI with conventional imaging sequences
and MR angiography. Clinically informative MR scanning can be performed in about 20 minutes, which is tolerated
by majority of the patients with acute stroke. This robust method is and obviously will be harvested in clinical
trials studying medical treatment of acute ischemic stroke. In the future, it is likely that combined DWI and PWI
is part of our clinical routine when examining patients with acute ischemic stroke.
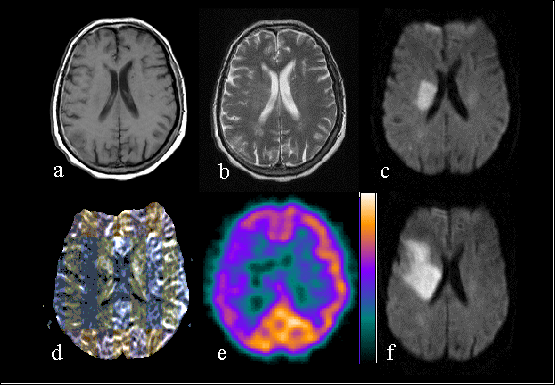
Figure 2. Seventy-three years old female with left hemiparesis. The first MRI (a-d) was performed
6.5 hours from the onset of symptoms. T1-weighted (a) and T2-weighted (b) image show very subtle findings. DWI
(C) clearly shows ischemic lesion on the right hemisphere. Map of relative cerebral blood flow (d) shows larger
perfusion defect on the right hemisphere, which is of about the same size than the perfusion defect on SPECT (e).
DWI on the next day shows the enlargement of the infarct.
3.2 Clinical applications: Tumor imaging
Blood volume mapping has been shown to be potentially useful in the grading of primary gliomas. MR blood volume
mapping was found to correlate strongly with PET-fluorodeoxyglucose (FDG) finding in most gliomas studied by Aronen
et al . They found that high-grade gliomas were more heterogeneous in blood volume maps compared with low-grade
lesions, making possible the grading of gliomas with CBV-mapping . Recently, it has been further confirmed that
MR perfusion imaging offers powerful tools for addressing pathophysiological changes associated with blood-tumor-barrier.
4. Brain activation studies
The development of fast magnetic resonance imaging methods have enabled monitoring of dynamic processes in the
human body. As long as one hundred years ago Roy and Sherrington introduced the idea of a close connection between
cerebral activity and cerebral circulation and since the production of the first useful MR brain images 1973, a
number of researchers have speculated on the possibility of imaging brain physiology as well as brain anatomy.
Recent development of ultrafast magnetic resonance imaging (MRI) has enabled a fast and accurate, non-invasive
way of human brain mapping - blood oxygenation level dependent contrast functional MRI (BOLD-fMRI) .
It has been known for a long time that the magnetic state of hemoglobin in red cells is strongly dependent on
oxygen saturation and that deoxygenated blood is considerably more paramagnetic than oxygenated blood. It is also
known that cortical neuronal activation not only increases regional oxygen consumption, but also regional blood
flow and regional blood volume. The sum of these events is a local transient hyperoxemia associated with neuronal
activity. This decrease of capillary and venous deoxyhemoglobin concentrations is reflected as an increase in the
MRI relaxation parameter T2* and consequently an increase in the signal intensity of T2* (susceptibility)-weighted
MR images. However, the signal changes related to cerebral activation are close to the noise level and therefore
numerous signal processing techniques are used to overcome the low signal-to-noise ratio . It was more than fifty
years later after the discovery of Pauling and Coryell until the first fMRI results were published . Since then
the BOLD technique has been successfully applied to many examinations of human visual, motor, somatosensory, auditory
and speech areas as well as in studies of higher cognitive functions.
4.1 Preoperative surgical planning
Until recent years non-invasive methods for determining individual functional anatomy prior to neurosurgical
procedures have not been available. Advances in several medical imaging modalities have made this possible. Today
it is possible to use positron emission tomography, functional MRI, magnetoencephalography (MEG), electroencephalography
(EEG), and transcranial magnetic stimulation to map eloquent brain areas before surgical intervention. Knowledge
of the location of motor areas or language areas, for example, will reduce the risk of iatrogenic damage causing
severe disability to the patient. This has also large economic impact as postoperative rehabilitation is expensive.
With functional MRI it possible to locate primary sensory and motor areas (Figure 3) as well as areas related
to higher cognitive functions such as language and memory and possibly also evaluate the hemispheric lateralization
of these functions Intra-operative real-time monitoring with functional MRI has now been demonstrated . Recent
results indicate that fMRI may also have a role in localization of epileptogenic foci . The studies evaluating
functional MRI in clinical use have so far been conducted on relatively small patient groups (10-20 patients).
The results have been encouraging in all studies performed.
Two main concerns in clinical use of fMRI are the artefacts and noise from motion and non-specificity to microvasculature.
Since patient movements of the order of couple of millimeters during the imaging can severely deteriorate the quality
of the data and induce severe artefacts, it is obvious that very good co-operation is required.
Other important movement related noise sources are the brain pulsation during the cardiac and respiratory cycles.
In the future, prospective motion compensation techniques may improve the clinical applicability to less co-operative
patient groups, such as children.
Currently the most widely used imaging sequences in fMRI are gradient-echo sequences, which provide the greatest
sensitivity to BOLD signal. They are, however, not selective to signals from microvasculature only. According to
optical imaging results the spread of signal to draining venules and thus introducing localization inaccuracy .
All forms of functional MRI can benefit from use of higher field strengths as signal to noise ratio increases linearly
with the field strength. This can be used for either shortening imaging times, higher resolution or increased sensitivity.
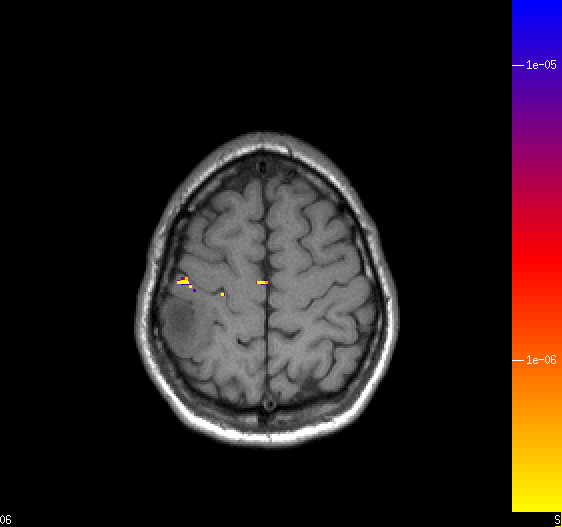
Figure 3. Sensorimotor cortex localized preoperatively in a tumor patient. The tumor is located
parietally immeaditely posterior to the primary sensory cortex. Resection could be performed without loss of sensorimotor
function.
4.2 Working memory and attention
Functional magnetic resonance imaging faces its greatest challenges and expectations in the field of cognitive
neuroscience. It is hoped that this method would lead to a deeper understanding of the neurophysiological basis
of behavior.
We have recently initiated a series of studies concerning neuronal mechanisms of visual and auditory working
memory and attention. Working memory refers to temporary storage of information that is processed in a range of
cognitive tasks . We applied the functional MR imaging technique to study the distribution of brain activation
in humans engaged in a visuospatial working memory task performance. Neurologically healthy subjects performed
a visuospatial n-back task with three load levels (0-, 1-, and 2-back tasks). In all subjects studied, significantly
activated voxels were detected in areas which - according to their Talairach coordinates - were located in the
middle frontal gyrus in the prefrontal cortex, in the superior frontal sulcus and gyrus and in the intraparietal
sulcus (Figure 4) . Several other cortical areas, including areas in the cingulate cortex, the insula, and the
superior and inferior parietal lobuli were also activated in relation to the task performance suggesting that a
network of distributed cortical areas is engaged in visuospatial working memory task performance in humans.
While visual working memory mechanisms have been studied extensively during the past years, the functional organization
of auditory working memory has received much less attention. There may be practical reasons for the lack of fMRI
studies on auditory working memory. Functional MR imaging is accompanied with a relatively high level of background
noise, which in auditory memory tasks may be a greater problem than when imaging during visual memory tasks. It
is, however, possible to trigger the imaging so that the background noise related to the imaging does not coincide
with the presentation of the auditory stimuli. On the other hand, the unavoidable background noise is similar in
all imaging conditions, minimizing its effects on the task-related changes in regional cerebral blood flow detected
by the fMRI. We have recently initiated an auditory fMRI project in which the functional organization of auditory
working memory is studied. By using the same n-back task paradigm as in our visual working memory task and by triggering
the imaging right after the end of the auditory stimulus, it was possible to overcome some of the problems related
to the background noise .
The visuospatial, as well as the auditive working memory task performance revealed marked consistency between
different subjects in the areas that were found to be statistically significantly activated during functional MR
imaging. The n-back task paradigm is thus well suited for functional MR imaging and may therefore prove diagnostically
useful in studying patients suffering from different kinds of memory disorders (e.g. dementias, Parkinsons disease,
stroke).
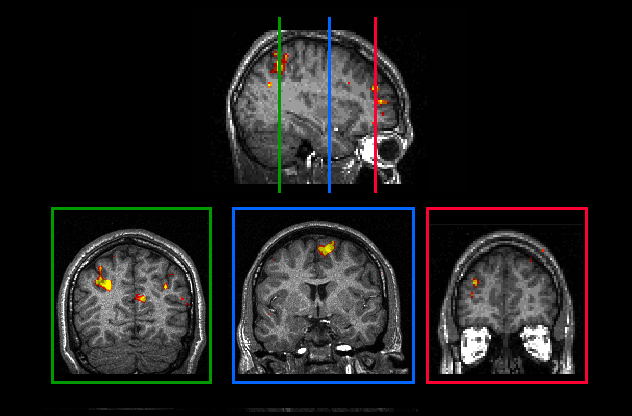
Figure 4. Visuospatial working memory as revealed by fMRI: A sagittal and three coronal slices
illustrating brain activation in the Medial Frontal Gyrus, Superior Frontal Sulcus, Superior Frontal Gyrus and
Intraparietal Sulcus.
5. Future Trends
Functional magnetic resonance imaging and magnetoencephalography provide a partial, but not overlapping view
of the physiological events. This implies that the combined use of these techniques, multimodal imaging, could
lead into a better understanding of human physiology . Most immediate applications of multimodal imaging are in
neurosurgery where mapping of brain functions can be used in the planning of operations and risk assessment as
well as intra-operatively for navigation in order to avoid unnecessary iatrogenic damage to the patient. Other
promising areas include diagnostics and characterization of many neurological conditions (especially ischemic strokes,
epilepsy and dementia) and psychiatric disorders.
Acknowledgements
Cancer Organization of Finland, Vuorisalo Foundation, Paavo Nurmi Foundation, Helsinki University Central Hospital
Research grant, Kuopio University Hospital Research Grant, the Academy of Finland, University of Helsinki grant
(no. 1127/62/96), Finnish Cultural Foundation, the Radiological Society of Finland, and the Pehr Oscar Klingendahl
Foundation
References
[1] Mansfield P: Multi-planar image formation using NMR spin echoes, Journal of Physics C, 10:L55-L58, 1977.
[2] Villringer A, Rosen BR, Belliveau JW, Ackerman JL, Lauffer RB, Buxton RB, Chao YS, Wedeen VJ, Brady TJ: Dynamic
imaging with lanthanide chelates in normal brain: contrast due to magnetic susceptibility effects, Magnetic Resonance
in Medicine, 6(2):164-174, 1988.
[3] Stejskal EO, Tanner JE: Spin diffusion measurements: spin echoes in the presence of a time-dependent field
gradient, J Chem Phys, 42(1):288-292, 1965.
[4] Tanner JE, Stejskal EO: Restricted self-diffusion of protons in colloidal systems by the pulsed-gradient, spin-echo
method, J Chem Phys, 49(4):1768-1777, 1968.
[5] Shimony JS, Akbudak E, McKinstry RC, Lori N, Cull TS, Conturo TE: Quantitative diffusion anisotropy imaging:
Normal values, anatomical findings, and preliminary results in relapsing remitting multiple sclerosis, Radiology,
209(P):240, 1998.
[6] Moseley ME, Cohen Y, Mintorovitch J, Chileuitt L, Shimizu H, Kucharczyk J, Wendland MF, Weinstein PR: Early
detection of regional cerebral ischemia in cats: comparison of diffusion- and T2-weighted MRI and spectroscopy,
Magnetic Resonance in Medicine, 14(2):330-346, 1990.
[7] Reith W, Hasegawa Y, Latour LL, Dardzinski BJ, Sotak CH, Fisher M: Multislice diffusion mapping for 3-D evolution
of cerebral ischemia in a rat stroke model, Neurology, 45(1):172-177, 1995.
[8] Hoehn-Berlage M, Norris DG, Kohno K, Mies G, Leibfritz D, Hossmann KA: Evolution of regional changes in apparent
diffusion coefficient during focal ischemia of rat brain: the relationship of quantitative diffusion NMR imaging
to reduction in cerebral blood flow and metabolic disturbances, Journal of Cerebral Blood Flow and Metabolism,
15(6):1002-1011, 1995.
[9] Marks MP, de Crespigny A, Lentz D, Enzmann DR, Albers GW, Moseley ME: Acute and chronic stroke: navigated spin-echo
diffusion-weighted MR imaging [published erratum appears in Radiology 1996 Jul;200(1):289], Radiology, 199(2):403-408,
1996.
[10] Sorensen AG, Buonanno FS, Gonzalez RG, Schwamm LH, Lev MH, Huang-Hellinger FR, Reese TG, Weisskoff RM, Davis
TL, Suwanwela N, Can U, Moreira JA, Copen WA, Look RB, Finklestein SP, Rosen BR, Koroshetz WJ: Hyperacute stroke:
evaluation with combined multisection diffusion- weighted and hemodynamically weighted echo-planar MR imaging,
Radiology, 199(2):391-401, 1996.
[11] Warach S, Chien D, Li W, Ronthal M, Edelman RR: Fast magnetic resonance diffusion-weighted imaging of acute
human stroke [published erratum appears in Neurology 1992 Nov;42(11):2192], Neurology, 42(9):1717-1723, 1992.
[12] Warach S, Gaa J, Siewert B, Wielopolski P, Edelman RR: Acute human stroke studied by whole brain echo planar
diffusion weighted magnetic resonance imaging, Annals of Neurology, 37(2):231-241, 1995.
[13] Lutsep HL, Albers GW, DeCrespigny A, Kamat GN, Marks MP, Moseley ME: Clinical utility of diffusion weighted
magnetic resonance imaging in the assessment of ischemic stroke, Annals of Neurology, 41(5):574-580, 1997.
[14] Lovblad KO, Baird AE, Schlaug G, Benfield A, Siewert B, Voetsch B, Connor A, Burzynski C, Edelman RR, Warach
S: Ischemic lesion volumes in acute stroke by diffusion-weighted magnetic resonance imaging correlate with clinical
outcome, Annals of Neurology, 42(2):164-170, 1997.
[15] Karonen J, Vainio P, Partanen K, Vanninen R, Korhonen K, Liu Y, Nuutinen J, Roivainen R, Onatsu J, Sivenius
J, Vanninen E, Könönen M, Kuikka J, Aronen H: Diffusion and perfusion MR imaging in the evaluation of
ischemic stroke during the first week. In: International society for magnetic resonance in medicine. Sixth scientific
meeting and exhibition, Sydney, Australia, 1998
[16] Rosen BR, Belliveau JW, Chien D: Perfusion imaging by nuclear magnetic resonance, Magnetic Resonance Quarterly,
5(4):263-281, 1989.
[17] Rosen BR, Belliveau JW, Vevea JM, Brady TJ: Perfusion imaging with NMR contrast agents, Magnetic Resonance
in Medicine, 14(2):249-265, 1990.
[18] Boxerman JL, Hamberg LM, Rosen BR, Weisskoff RM: MR contrast due to intravascular magnetic susceptibility perturbations,
Magnetic Resonance in Medicine, 34(4):555-566, 1995.
[19] Fisel CR, Ackerman JL, Buxton RB, Garrido L, Belliveau JW, Rosen BR, Brady TJ: MR contrast due to microscopically
heterogeneous magnetic susceptibility: numerical simulations and applications to cerebral physiology, Magnetic
Resonance in Medicine, 17(2):336-347, 1991.
[20] Weisskoff RM, Zuo CS, Boxerman JL, Rosen BR: Microscopic susceptibility variation and transverse relaxation:
theory and experiment, Magnetic Resonance in Medicine, 31(6):601-610, 1994.
[21] Ostergaard L, Sorensen AG, Kwong KK, Weisskoff RM, Gyldensted C, Rosen BR: High resolution measurement of cerebral
blood flow using intravascular tracer bolus passages. Part II: Experimental comparison and preliminary results,
Magnetic Resonance in Medicine, 36(5):726-736, 1996.
[22] Ostergaard L, Weisskoff RM, Chesler DA, Gyldensted C, Rosen BR: High resolution measurement of cerebral blood
flow using intravascular tracer bolus passages. Part I: Mathematical approach and statistical analysis, Magnetic
Resonance in Medicine, 36(5):715-725, 1996.
[23] Stewart GN: Researches on the circulation time in organs and on the influences which affect it. Parts I-III.,
J Physiol (London), 15:1, 1894.
[24] Ostergaard L, Smith DF, Vestergaard-Poulsen P, Hansen SB, Gee AD, Gjedde A, Gyldensted C: Absolute cerebral
blood flow and blood volume measured by magnetic resonance imaging bolus tracking: comparison with positron emission
tomography values, Journal of Cerebral Blood Flow and Metabolism, 18(4):425-432, 1998.
[25] Warach S, Dashe JF, Edelman RR: Clinical outcome in ischemic stroke predicted by early diffusion- and perfusion
weighted magnetic resonance imaging: a preliminary analysis, Journal of Cerebral Blood Flow and Metabolism, 16(1):53-59,
1996.
[26] Baird AE, Benfield A, Schlaug G, Siewert B, Lovblad KO, Edelman RR, Warach S: Enlargement of human cerebral
ischemic lesion volumes measured by diffusion-weighted magnetic resonance imaging, Annals of Neurology, 41(5):581-589,
1997.
[27] Tong DC, Yenari MA, Albers GW, O'Brien M, Marks MP, Moseley ME: Correlation of perfusion- and diffusion-weighted
MRI with NIHSS score in acute (<6.5 hour) ischemic stroke, Neurology, 50(4):864-870, 1998.
[28] Barber PA, Darby DG, Desmond PM, Yang Q, Gerraty RP, Jolley D, Donnan GA, Tress BM, Davis SM: Prediction of
stroke outcome with echo planar perfusion and diffusion weighted MRI, Neurology, 51(2):418-426, 1998.
[29] Karonen JO, Kuikka J, Ostergaard L, Vainio P, Vanninen R, Aronen HJ: Comparison of MR diffusion and perfusion
imaging to SPECT in acute ischemic stroke during the first week. In: Radiological Society of North America, 84th
Scientific Scientific Assembly and Annual Meeting, Chicago, USA, 1998
[30] Sorensen AG, Copen WA, Ostergaard L, Buonanno FS, Gonzalez RG, Rordorf G, Rosen BR, Schwamm LH, Weisskoff RM,
Koroshetz WJ: Hyperacute stroke: simultaneous measurement of relative cerebral blood volume, relative cerebral
blood flow and mean transit time, Radiology, 210:519-527, 1999.
[31] Karonen JO, Nuutinen J, Vanninen R, Vainio P, Liu Y, Perkiö J, Korhonen K, Roivainen R, Partanen K, Sivenius
J, Vanninen E, Kuikka J, Ostergaard L, Aronen HJ: Combined diffusion and perfusion MRI in acute ishemic stroke:
correlation to clinical outcome. In: International society for magnetic resonance in medicine, Seventh scientific
meeting and exhibition, Philadelphia, Pennsylvania, USA, 1999
[32] Aronen HJ, Cohen MS, Belliveau JW, Fordham JA, Rosen BR: Ultrafast imaging of brain tumors, Topics in Magnetic
Resonance Imaging, 5(1):14-24, 1993.
[33] Aronen HJ, Gazit IE, Louis DN, Buchbinder BR, Pardo FS, Weisskoff RM, Harsh GR, Cosgrove GR, Halpern EF, Hochberg
FH, et al.: Cerebral blood volume maps of gliomas: comparison with tumor grade and histologic findings, Radiology,
191(1):41-51, 1994.
[34] Ostergaard L, Hochberg FH, Rabinov JD, Sorensen AG, Lef M, Kim L, Weisskoff RM, Gonzalez RG, Gyldensted C,
Rosen BR: Early changes measured by magnetic resonance imaging in cerebral blood flow, blood volume, and blood-brain
barrier permeability following dexamethasone treatment in patients with brain tumors, Journal of Neurosurgery,
90(2):300-305, 1999.
[35] Roy CS, Sherrington CS: On the regulation of the blood-supply of the brain, Journal of Physiology, 11:85-108,
1890.
[36] Ogawa S, Lee TM, Kay AR, Tank DW: Brain magnetic resonance imaging with contrast dependent on blood oxygenation,
Proceedings of the National Academy of Sciences of the United States of America, 87(24):9868-9872, 1990.
[37] Pauling L, Coryell CD: The magnetic properties and structure of hemoglobin, oxyhemoglobin, and carbonmonooxyhemoglobin,
Proceedings of the National Academy of Sciences of the United States of America, 22:210-216, 1936.
[38] Thulborn KR, Waterton JC, Matthews PM, Radda GK: Oxygenation dependence of the transverse relaxation time of
water protons in whole blood at high field, Biochim Biophys Acta, 714(2):265-270, 1982.
[39] Bandettini PA, Jesmanowicz A, Wong EC, Hyde JS: Processing strategies for time-course data sets in functional
MRI of the human brain, Magnetic Resonance in Medicine, 30(2):161-173, 1993.
[40] Friston KJ, Holmes AP, Poline JB, Grasby PJ, Williams SC, Frackowiak RS, Turner R: Analysis of fMRI time-series
revisited, Neuroimage, 2(1):45-53, 1995.
[41] Belliveau JW, Kennedy DN, Jr., McKinstry RC, Buchbinder BR, Weisskoff RM, Cohen MS, Vevea JM, Brady TJ, Rosen
BR: Functional mapping of the human visual cortex by magnetic resonance imaging, Science, 254(5032):716-719, 1991.
[42] Kwong KK, Belliveau JW, Chesler DA, Goldberg I E, Weisskoff RM, Poncelet BP, Kennedy DN, Hoppel BE, Cohen MS,
Turner R, et al: Dynamic magnetic resonance imaging of human brain activity during primary sensory stimulation,
Proceedings of the National Academy of Sciences of the United States of America, 89(12):5675-5679, 1992.
[43] Morioka T, Mizushima A, Yamamoto T, Tobimatsu S, Matsumoto S, Hasuo K, Fujii K, Fukui M: Functional mapping
of the sensorimotor cortex: combined use of magnetoencephalography, functional MRI, and motor evoked potentials,
Neuroradiology, 37(7):526-530, 1995.
[44] Puce A, Constable RT, Luby ML, McCarthy G, Nobre AC, Spencer DD, Gore JC, Allison T: Functional magnetic resonance
imaging of sensory and motor cortex: comparison with electrophysiological localization, Journal of Neurosurgery,
83(2):262-270, 1995.
[45] Pujol J, Conesa G, Deus J, Lopez-Obarrio L, Isamat F, Capdevila A: Clinical application of functional magnetic
resonance imaging in presurgical identification of the central sulcus, Journal of Neurosurgery, 88(5):863-869,
1998.
[46] Shimizu H, Nakasato N, Mizoi K, Yoshimoto T: Localizing the central sulcus by functional magnetic resonance
imaging and magnetoencephalography, Clinical Neurology & Neurosurgery, 99(4):235-238, 1997.
[47] Yetkin FZ, Mueller WM, Morris GL, McAuliffe TL, Ulmer JL, Cox RW, Daniels DL, Haughton VM: Functional MR activation
correlated with intraoperative cortical mapping, American Journal of Neuroradiology, 18(7):1311-1315, 1997.
[48] Yousry TA, Schmid UD, Schmidt D, Hagen T, Jassoy A, Reiser MF: The central sulcal vein: a landmark for identification
of the central sulcus using functional magnetic resonance imaging, Journal of Neurosurgery, 85(4):608-617, 1996.
[49] Korvenoja A, Huttunen J, Salli E, Pohjonen H, Martinkauppi S, Palva M, Sipilä L, Virtanen J, Ilmoniemi
RJ, Aronen HJ: The time course activation of multiple cortical areas by somatosensory stimulation: a model based
on combined magnetoencephalographic and functional magnetic resonance imaging, Accepted for publication in Human
Brain Mapping, In press,
[50] Desmond JE, Sum JM, Wagner AD, Demb JB, Shear PK, Glover GH, Gabrieli JD, Morrell MJ: Functional MRI measurement
of language lateralization in Wada-tested patients, Brain, 118:1411-1419, 1995.
[51] Detre JA, Maccotta L, King D, Alsop DC, Glosser G, M DE, Zarahn E, Aguirre GK, French JA: Functional MRI lateralization
of memory in temporal lobe epilepsy, Neurology, 50(4):926-932, 1998.
[52] Gering DT, Weber DM: Intraoperative, real-time, functional MRI, Journal of Magnetic Resonance Imaging, 8(1):254-257,
1998.
[53] Connelly A: Ictal imaging using functional magnetic resonance, Magnetic Resonance Imaging, 13(8):1233-1237,
1995.
[54] Detre JA, Alsop DC, Aguirre GK, Sperling MR: Coupling of cortical and thalamic ictal activity in human partial
epilepsy: demonstration by functional magnetic resonance imaging, Epilepsia, 37(7):657-661, 1996.
[55] Jackson GD, Connelly A, Cross JH, Gordon I, Gadian DG: Functional magnetic resonance imaging of focal seizures,
Neurology, 44(5):850-856, 1994.
[56] Warach S, Ives JR, Schlaug G, Patel MR, Darby DG, Thangaraj V, Edelman RR, Schomer DL: EEG-triggered echo-planar
functional MRI in epilepsy, Neurology, 47(1):89-93, 1996.
[57] Lee CC, Jack CR, Jr., Grimm RC, Rossman PJ, Felmlee JP, Ehman RL, Riederer SJ: Real-time adaptive motion correction
in functional MRI, Magnetic Resonance in Medicine, 36(3):436-444, 1996.
[58] Lee CC, Grimm RC, Manduca A, Felmlee JP, Ehman RL, Riederer SJ, Jack CR, Jr: A prospective approach to correct
for inter-image head rotation in fMRI, Magnetic Resonance in Medicine, 39(2):234-243, 1998.
[59] Menon RS, Ogawa S, Hu X, Strupp JP, Anderson P, Ugurbil K: BOLD based functional MRI at 4 Tesla includes a
capillary bed contribution: echo-planar imaging correlates with previous optical imaging using intrinsic signals,
Magnetic Resonance in Medicine, 33(3):453-459, 1995.
[60] Baddley A: Working memory, Clarendon Press, Oxford, 1986,
[61] Talairach J, Tournoux P: Co-planar stereotaxic atlas of the human brain, George Thieme Verlag, Stuttgart, 1988,
[62] Carlson S, Martinkauppi S, Rämä P, Salli E, Korvenoja A, Aronen HJ: Distribution of cortical activation
during visuospatial n-back tasks as revealed by functional magnetic resonance imaging, Cerebral Cortex, 8:743-752,
1998.
[63] Martinkauppi S, Rämä P, Korvenoja A, Aronen HJ, Carlson S: Functional Magnetic Resonance Imaging
During Audiospatial Working Memory Tasks with Different Memory Loads. In: Human Brain Mapping, Dusseldorf, Germany,
1999
[64] Ilmoniemi RJ, Aronen HJ: Moonen C and Bandettini PA, Eds. Cortical excitability and connectivity reflected
in fMRI, MEG, EEG, and TMS, Springer Verlag, 1999, 453-463

 Home
Current Issue
Table of Contents
Home
Current Issue
Table of Contents