|
Imaging of Interictal Epileptiform Discharges
Using Spike-triggered fMRI
K. Krakowa, P.J. Allenb, M.R. Symmsa, D.R. Fisha,b, and L. Lemieuxa
a Epilepsy Research Group, Dept. of Clinical Neurology, Institute of Neurology, UCL, London, UK
b Dept. of Clinical Neurophysiol., National Hospital for Neurology and Neurosurgery, London, UK
Abstract. EEG-triggered functional MRI (fMRI) offers the potential to localize the generators of scalp EEG events,
such as interictal epileptiform discharges, using a biological measurement as opposed to relying solely on modelling
techniques. We have addressed and resolved the issues of patient safety and EEG quality inside the MR scanner and
report here the application of this technique in 29 experiments in 14 patients with localization-related epilepsy
and frequent interictal epileptiform discharges (spikes or spike wave). In each experiment, 20-slice snapshot gradient
echo echo-planar images (EPI) were acquired approximately 3.5 seconds after single epileptiform discharges (activation
image) and in the absence of discharges (control image). Between 21 and 50 epileptiform discharges were sampled
in each experiment. The significance of functional activation was tested on a pixel-by-pixel basis using statistical
parametric mapping. Eight of the 14 patients showed focal changes of the blood oxygen level dependent (BOLD) signal,
which occurred in close spatial relation to the maximum of the epileptiform discharges in the concurrent EEG. Six
of the eight patient showing a fMRI activation were studied at least twice and had reproducible results. We conclude
that EEG-triggered fMRI is now a sufficiently developed technique to be more widely used in clinical studies, demonstrating
that it can reproducibly localize brain areas involved in the generation of spikes and spike wave in epilepsy patients
with frequent interictal discharges which is not possible with other non-invasive techniques.
1. Introduction
For more than 50 years interictal epileptiform discharges (IED) recorded by scalp EEG are the mainstay for diagnosing
and classifying the type of epilepsy. The knowledge of the underlying generators of these EEG events, however,
is still limited. Due to their restricted spatial sampling and the "inverse
problem", neither EEG or MEG (magnetoencephalography) can directly
identify these generators. On the other hand, the low temporal resolution of PET and SPECT prevents the investigation
of brain activation linked to brief IED.
In contrast it has been shown recently that functional MRI (fMRI) allows the detection of local changes in blood
oxygenation associated not only with epileptic seizures [1] but also IED [2,3,4]. The acquisition of MR images
linked to brief subclinical events like IED requires the recording of EEG during the MR scanning procedure [5,6].
We have previously reported on a fMRI- compatible EEG acquisition system that ensures patient safety (mainly through
current-limiting resistors) [7], excellent EEG quality (using on-line pulse artifact suppression) [8] and functional
images with minimum artifacts (by using appropriate materials for electrode assemblies and shielding of electromagnetic
noise) [9]. We have used this optimized recording technique to monitor the EEG of epilepsy patients undergoing
MRI and triggered ultra-fast snapshot multi-slice EPI blood oxygen level dependent (BOLD) fMRI acquisitions after
single IED (spike or spike wave) were identified in the on-line EEG [4]. The purpose of this study was to identify
the generators of the IED and correlate their site to the focus of previous interictal scalp EEG recordings, invasive
and ictal EEG recordings and, if present, structural lesions in the anatomical MRI.
2. Methods
2.1 Patients
Fourteen patients (10 male, 4 female, median age 27 years, range: 17 - 48) with a confirmed diagnosis of medically
intractable localization-related epilepsy were studied. The study was approved by the ethics committee of the National
Hospital for Neurology and Neurosurgery and all patients gave informed consent. Ten patients had lesions in the
structural MRI (5 x cortical dysgenesis, 2 x hippocampal sclerosis, low grade astrocytoma, posttraumatic brain
damage), the remaining 4 patients had a normal structural MRI. All patients showed frequent stereotyped focal epileptiform
discharges in previous routine 20 channel scalp EEG recordings with an average of at least one epileptiform discharge
per minute. Patients with less frequent or generalized IED were not included in the study.
2.2 EEG recording
EEG was recorded in the MR scanner using the following system: Standard Ag/AgCl disk electrodes were applied
on the scalp using collodium; these had 12 kOhm current limiting resistors fitted adjacent to each electrode [7].
The electrodes were connected to a non-ferrous headbox (developed in-house) placed at the entrance to the bore
of the magnet. The headbox was connected to a Neurolink Patient Module (Physiometrix, MA, USA) which digitizes
and transmits the EEG signal out of the scanner room via a fibre optic cable to the Neurolink Monitor Module, which
reconstructs the analog EEG signals. These were then recorded using a digital EEG recording system (sample rate
200 Hz, bandwidth: 0.12-50 Hz).
For each experiment, 12 electrodes were applied to the scalp positions FP1/FP2, F7/F8, T3/T4, T5/T6, O1/O2,
Fz and Pz according to the 10/20 system. In addition, two precordial ECG channels were recorded to facilitate pulse
artifact subtraction (75 kOhm current limiting resistors were fitted to each ECG-electrode) [8]. EEG data was digitally
remontaged and displayed to show bitemporal chains. In 12 patients, on-line pulse artifact subtraction software
was used to aid visual detection of the epileptiform discharges. This method subtracts an averaged pulse artifact
waveform calculated for each electrode during the previous 10 seconds. Technical details have been described elsewhere
[8].
2.3 fMRI acquisition and processing
FMRI was performed on a 1.5 T Horizon EchoSpeed MRI scanner (General Electric, Milwaukee, USA) using snapshot
gradient-echo EPI (TE = 40 ms, 24 cm field-of-view). Acquisitions of 20 contiguous 5 mm slices with a 64x64 matrix
were performed at each time-point. The acquisition time was 4.5 seconds. Additional high resolution multi-shot
EPI images (matrix 256x256, 16 shots, TR = 3 s, all other parameters as fMRI data) were acquired. These images
have geometric distortions similar to the fMRI data and were used as anatomical references for the fMRI data.
Images were acquired after "activation-"and "control-" states, defined by visual inspection of the on-line EEG.
The activation state was defined as a single stereotyped
IED (spike or spike wave). As the peak blood oxygenation level change detected by fMRI occurs approximately 4 to
7 seconds after the onset of the brain activity [10,11], a delay of approximately 3.5 seconds between the observation
of the discharge and the image acquisition was applied. Control images were acquired after periods of at least
10 seconds of background EEG activity without epileptiform activity. Image acquisition was performed non-periodically
with activation and control images interleaved, depending on the sequence of the EEG events. An interval of at
least 15 seconds was established between successive acquisitions to ensure the same T1-weighting for each acquisition.
Due to hardware restrictions, the number of time-points was limited to 98 per study. This led to a maximum acquisition
of 49 activation- and control- states, respectively, as equal numbers of activation and control states were used
for the statistical analysis. The typical total scanning time was 60 to 90 minutes, depending the frequency of
EEG events. The SPM96 package was used to perform spatial realignment and statistical analysis [12]. The significance
threshold was set to p < 0.001 and the extent threshold to p < 0.05.
3. Results
In all 29 experiments the EEG-quality was sufficient to detect activation and control periods reliably throughout
the study, though in 24 experiments (twelve of the patients) on-line pulse artifact subtraction was necessary to
achieve good EEG quality. IED recorded inside the scanner had a similar localisation, amplitude and configuration
as in previous recordings under routine conditions. None of the patients reported discomfort or other adverse events
due to the EEG recording during the experiments. Between 21 and 49 fMRI acquisitions triggered after IED were sampled
in each study, the smallest number of IED leading to a significant fMRI activation was 34. In most of the experiments
sing a positive result, 45 to 49 activation timepoints were sampled.
In 8 out of 14 patients, a focal activation was seen in the fMRI data. In all cases this was a single cortical
area. Four of the fMRI positive patients had a cortical dysgenesis, one a hippocampal sclerosis and one a low grade
astrocytoma; two patients had a normal structural MRI. Figure 1 presents the SPM96 activation map of a patient
with hippocampal sclerosis, showing an activation in the mesial temporal lobe. In all patients, the fMRI activation
showed co-localization with the EEG spike-focus; in all patients with lesional epilepsy, the fMRI was overlapping
or adjacent to the lesion. Six of the 8 patients underwent repeated (2 to 5) experiments, in all of these cases
the activation was reproducible.
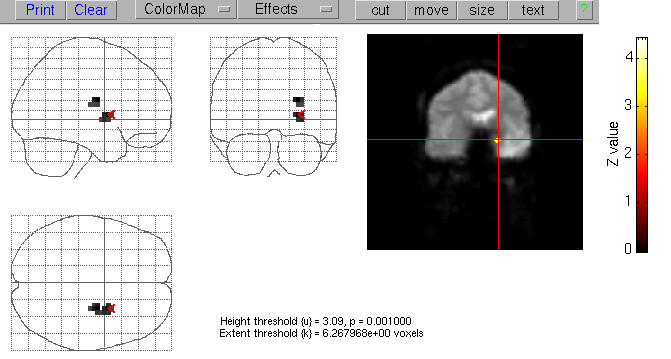
Figure 1. SPM activation map showing activation of the left mesial temporal lobe.
Discussion
In this study we were able to obtain a good quality EEG in the MR scanner in all experiments, detect spontaneous
IED on-line and trigger EPI BOLD acquisitions after these events. The MR image quality was not significantly compromised
by the EEG recording and in 8 out of 14 patients we found focal MR signal increases associated with the focal IED
seen in the concurrent EEG. These activations showed co-localisation with the focus seen in previous routine scalp
EEG and the EEG recording during the experiment. Additional electrocorticography was performed in one patient and
confirmed the co-localization between interictal epileptiform activity and fMRI activation. In the four patients
with fMRI activation who had previous lateralizing ictal EEG recordings, the lateralization was concordant. Activation
was seen in patients with different underlying pathologies (chronic encephalitis, hippocampal sclerosis, cortical
dysgenesis, and tumour). Figure 1 shows the activation map of a patient with refractory left mesial temporal lobe
seizures who underwent previous epilepsy surgery with a left anterior temporal lobectomy without improving the
seizure frequency. The fMRI revealed an activation in the remaining (sclerotic) mesial temporal lobe, in keeping
with an epileptogenic zone beyond the previous resection. The activation of deep temporal structures is remarkable
as it is correlated with epileptiform discharges recorded with scalp EEG. This requires propagation of the epileptiform
activity to a larger superficial cortical area. An activation solely in deep structures might suggest that fMRI
more readily identified the site of primary spike generation. This hypothesis would be in keeping with the result
of a patient with bilateral occipital discharges, who showed a unilateral occipital fMRI activation on the side
of the EEG predominance. The possibility that the site of the primary generator of epileptic activity could be
associated with different metabolic and hemodynamic changes compared to brain areas involved in the propagation
of this activity requires further studies given its potential clinical relevance.
4.1. Methodological Considerations
Investigations of epileptic foci in humans have been hitherto limited by either the low spatial or temporal
resolution of the available diagnostic tools. Due to their restricted spatial sampling and the insolvable "inverse problem"
of working back from distant scalp potentials to hypothesise about the likely sites of their generators, neither
EEG nor MEG can directly localize the source of epileptic activity. PET and SPECT studies have shown an increased
blood flow and metabolism in the region of the seizure focus during ictal events [13,14] and, in contrast, a decreased
blood flow and metabolism during the interictal state [15]. Due to their low temporal resolution, however, these
methods sample activity continuously over a prolonged period of time, and hence cannot investigate the changes
in blood flow and oxygenation related to brief IED. By time-locking the fMRI acquisition to single EEG events,
we could confirm results of previous case reports [2,3,16] that EEG-triggered fMRI is a practicable method to identify
brain activation associated with subclinical discharges with a high spatial resolution and completely non-invasively
[4].
Methodological limitations of spike-triggered fMRI are caused by genuine BOLD imaging characteristics. Firstly,
the BOLD contrast signal changes after a brief neuronal activation start to increase approximately 2 seconds after
the stimulus, peak after 4 to 7 seconds and last about 10 seconds with a high variability [10,11]. Thus, these
underlying hemodynamics prevent the distinction between sources sequentially activated within a few seconds or
even fractions of a second during propagation of IED. Secondly, the low signal to noise ratio of BOLD imaging requires
sampling of activation and control states. Using a 1.5 Tesla scanner, we found that at least 30 IED had to be sampled
to obtain an activation clearly distinguishable from noise, even when low resolution images (64x64 matrix), with
a relatively high signal to noise ratio (-100), were used. This limits the practicability
of this method to patients with frequent IED and the duration of the study is highly dependent on the frequency
of appropriate EEG events. There is a trade off, whereby increasing the number of events sampled improves signal
to noise but requires a prohibitive scanning time and may increase misregistration problems caused by patient movement.
As IED occur unpredictably, the MRI data were acquired in a non-periodic manner with an interleaved sampling
of activation and control states. This type of non-periodic acquisition requires an interval of about 15 seconds
between acquisitions to allow the NMR spins to return to equilibrium. Data collection efficiency is thus in the
range of only 10%. Furthermore, in contrast to continuous image acquisition EEG-triggered fMRI requires on-line
analysis of the EEG. An advantage of spike-triggered fMRI is the possibility to maintain the desired activation
to control image ratio which substantially reduces the number of acquisition periods required.
It remains unclear why 6 of the studied epilepsy patients did not show a fMRI activation. While EEG-triggered
fMRI offers the possibility of a specific detection of local blood oxygenation changes associated with interictal
epileptiform discharges, it might not be sensitive enough to detect all activated areas. Further improvements of
the signal to noise ratio are therefore required. The applied thresholding in particular may also account for the
relatively small size of the fMRI activation compared to the cortical areas involved in generating epileptiform
discharges found by electrocorticography.
4.2. Clinical Relevance
Functional (perfusion [17,18], diffusion [19], and BOLD [1,20] ) MRI studies can detect focal MR signal changes
associated with ictal activity. However, ictal MR scanning is for practical reasons limited to patients with predictable
seizure (e.g. reflex epilepsy), seizure series, or continuous seizure activity and is likely to be compromised
by seizure-related movement artifacts. Furthermore, functional imaging of ongoing epileptic seizures is likely
to be confounded by propagation of the ictal activity. With the possibility of recording EEG inside the MR scanner,
IED can be detected during the scanning session and can be used to trigger scan acquisitions [4]. This approach
is likely to find a broader application than ictal fMRI, because IED are present in most of epilepsy patients and
do not have clinical correlates, in particular motion, which can compromise fMRI quality.
Knowledge of the generators of interictal events identified by EEG-triggered fMRI would provide crucial information
for :
interpreting the findings of routine EEG studies. The detection of interictal epileptiform discharges in the
scalp EEG has been the mainstay for the diagnosis and classification of epilepsy for more than fifty years. However,
the EEG interpretation is still limited by the impossibility to identify directly the underlying generators of
EEG events. This is due to the restricted spatial sampling and the hitherto insoluble Ainverse
problem@ of working back from distant scalp potentials to hypothesise
about the likely sites of their generators. The distribution of fMRI-derived cortical activation could be used
to constrain generator modelling of the scalp-recorded epileptiform discharges and thereby may be helpful in addressing
the inverse problem which limits the interpretation of scalp EEG;
understanding the underlying pathophysiological mechanisms of epilepsy, e.g. the neuro-vascular coupling of
epileptic activity;
relating the anatomical site of the underlying structural abnormalities to the sites of functional disturbance;
planning the appropriate extent of surgical resection in respect of different lesional pathologies in pharmaco-resistant
patients undergoing epilepsy surgery. Experimental work has indicated that there are likely to be different mechanisms
of epileptogenesis, and outcome of epilepsy surgery appears to be crucially related to pathology [21].
The main diagnostic question in the presurgical evaluation of epilepsy patients is to localize the area of brain
necessary to generate seizures, the "epileptogenic zone". fMRI triggered after interictal epileptiform discharges localises
brain areas being involved in generating these particular EEG events. The area of cortex that generates interictal
spikes is labelled as "irritative zone".
This is not necessarily identical with the "epileptogenic zone", but has typically a close spatial relationship with it [22,23].
Hence, the localization of brain areas contributing to the irritative zone by fMRI has the potential to become
a useful additional non-invasive method in the presurgical evaluation of patients with intractable epilepsy. To
determine the significance of EEG/fMRI findings, further work is needed to compare the results to the anatomical
extent of the spiking cortex identified by electrocorticography and to the surgical outcome in relation to the
extent of removal of the activated area in those patients subsequently undergoing epilepsy surgery.
5. Conclusion
EEG-triggered fMRI can identify brain areas involved in generating interictal epileptiform discharges with a
high spatial resolution. This non-invasive method has the potential to improve the understanding of the pathophysiology
of epilepsy, interpretation of scalp EEG findings, and assist in the presurgical evaluation of patients with intractable
partial seizures.
Acknowledgements
This study was partly funded by the Medical Research Council, UK.
References
[1] Jackson GD, Connelly A, Cross JH, Gordon I, Gardian DG: Functional magnetic resonance imaging of focal seizures.
Neurology 44:850-856,1994.
[2] Warach S, Ives JR, Schlaug G, Patel MR, Darby DG, Thangaraj V, Edelman RR, Schomer DL: EEG-triggered echo-planar
functional MRI in epilepsy. Neurology 47:89-93,1996.
[3] Seeck M, Lazeyras F, Michel CM, Blanke O, Gericke CA, Ives J, Delavelle J, Golay X, Haenggeli CA, de Tribolte
N, Landis T: Non-invasive epileptic focus localization using EEG-triggered functional MRI and electromagnetic tomography.
EEG Clin Neurophys 106:508-512,1998.
[4] Krakow K, Woermann FG, Symms MR, Allen PJ, Lemieux L, Barker GJ, JS Duncan, Fish DR: EEG-triggered functional
MRI of interictal epileptiform activity in patients with partial seizures. Brain 1999 (in press).
[5] Ives JR, Warach S, Schmitt F, Edelman RR, Schomer DL: Monitoring the patient=s EEG during echo planar MRI.
Electroenceph Clin Neurophys 87:417-420,1993.
[6] Huang-Hellinger FR, Breiter HC, McCormack G, Cohen MS, Kwong KK, Sutton JP, Savoy RL, Weisskoff RM, Davis TL,
Baker JR, Belliveau JW, Rosen BR: Simultaneous functional magnetic resonance imaging and electrophysiological recording.
Human Brain Mapping 3:13-23,1995.
[7] Lemieux L, Allen PJ, Franconi F, Symms MR, Fish DR: Recording of EEG during fMRI experiments: patient safety.
Magn Reson in Med 38:943-952,1997.
[8] Allen PJ, Polizzi G, Krakow K, Fish DR, Lemieux L: Identification of EEG events in the MR scanner: the problem
of pulse artifact and a method for its subtraction. Neuroimage 8:229-239,1998.
[9] Krakow K, Allen PJ, Symms MR, Lemieux L, Fish DR: The effect of EEG recording on fuctional image quality. Proceedings
of the International Society for Magnetic Resonance in Medicine, Philadelphia, 1999 (in press).
[10] Hennig J, Janz C, Speck O, Ernst T: Functional spectroscopy of brain activation following a single light impulse:
examinations of the mechanisms of fast initial response. International Journal of Imaging Systems and Technology
6:203-208,1995.
[11] Rosen BR, Buckner RL, Dale AM: Event-related functional MRI: past, present, and future. Proc Natl Acad Sci
USA 95:773-780,1998.
[12] Friston KJ, Holmes AP, Worsley KJ, Poline JB, Frith CD, Frackowiak RSJ: Statistical parametricmaps in functional
imaging: A general linear approach. Human Brain Mapping 2:189-210,1995.
[13] Engel JJ, Kuhl DE, Phelps ME, Rausch R, Nuwer M: Local cerebral metabolism during partial seizures. Neurology
33:400-413,1983.
[14] Lee BI, Markand ON, Siddiqui AR, Park HM, Mock B, Wellman HH, Worth RM, Edwards MK: Single photon emission
computed tomography (SPECT) brain imaging using N,N,N=-trimethyl-N=-(2 hydroxy-3-methyl-5-123I-iodobenzyl)-1,3-propanediamine
2 HCl (HIPDM): intractable complex partial seizures. Neurology 36:1471-1476,1986.
[15] Engel JJ, Kuhl DE, Phelps DE, Crandall PH: Comparative localization of epileptic foci in partial epilepsy by
PCT and EEG. Ann Neurol 12: 529-537,1982.
[16] Symms MR, Allen PJ, Woermann FG, Polizzi G, Krakow K, Barker GJ, Fish DR, Duncan JS: Reproducible localisation
of interictal epileptiform discharges using EEG correlated fMRI. Proceedings of the International Society for Magnetic
Resonance in Medicine, Sydney, 1998: 168.
[17] Fish DR, Brooks DJ, Young IR, Bydder GM: Use of magnetic resonance imaging to identify changes in cerebral
blood flow in epilepsia partialis continua. Magn Reson Med 8:238-240,1988.
[18] Warach S, Levin JM, Schomer DL, Holman BL, Edelman RR: Hyperperfusion of ictal seizure focus demonstrated by
MR perfusion imaging. AJNR 15:965-968,1994.
[19] Wieshmann UC, Symms MR, Shorvon SD: Diffusion changes in status epilepticus. Lancet 350:493-494,1997.
[20] Detre JA, Sirven JI, Alsop DC, O´Connor MJ, French JA: Localization of subclinical ictal activity by
functional magnetic resonance imaging: Correlation with invasive monitoring. Ann Neurol 38:618-624,1995.
[21] Engel JJ, Pedley TA, eds: Epilepsy. A comprehensive textbook. Philadelphia: Lippincott-Raven,1998; Vol. I,
Chapter 79, 919-935.
[22] Ebersole JS: EEG and MEG dipole source modeling. In: Engel JJ, Pedley TA, editors. Epilepsy. A comprehensive
textbook. Lippincott-Raven, Philadelphia, 1998: 919-935.
[23] Lüders HO, Engel JJ, Munari C: General principles. In: Engel JJ, editor. Surgical treatment of the epilepsies.
Raven Press, New York, 1993: 137-153.
|

 Home
Current Issue
Table of Contents
Home
Current Issue
Table of Contents

