Correspondence: Mr S A M
Nashef, Papworth Hospital, Papworth Everard, Cambridge,
UK CB3 8RE.
E-mail: sam.nashef@euroscore.org,
phone +44 1480 364299, fax +44 1480 364744
1. Introduction
1.1. What is Cardiac Output?
The cardiac output is the amount of blood
pumped to the peripheral circulation by the heart every
minute. It is a measurement that reflects the status of
the entire circulatory system, not just the heart, since
it is governed by autoregulation and the metabolic demands
of the tissues.
Cardiac output (CO) is thus defined as
the amount of blood ejected from the ventricle per minute,
and is measured in litres per minute (L/min).
1.2. Why Monitor the Cardiac Output?
The care of many critically ill or unstable
patients crucially depends on adequate perfusion of tissues
and organs with oxygenated blood. The availability and adequacy
of oxygenation of the circulating arterial blood is easy
to measure. However, the adequacy of delivery of this blood
to the peripheral tissues by the pump action of the heart,
i.e. the CO, is more difficult to assess accurately.
Clinical indicators provide only an indirect
measure of CO, and rely on the skill and experience of the
clinician. When using clinical indicators to determine whether
the CO is satisfactory or not, they need to be taken as
a group and used with cautious extrapolation. Some of these
indicators are:
1. skin temperature: if the patient is warm, the CO
is probably not abnormally reduced, as the skin is one of
the first organs to shut down in low CO states.
2. urinary output: if urine output is normal, this
suggests adequate perfusion of the kidneys and that the
CO is probably not drastically low.
3. blood pressure: mean arterial pressure (MAP) is
very easily measured and correlates well with CO provided
systemic vascular resistance (SVR) remains constant. (MAP=
CO x SVR)
Unfortunately, these assumptions, usually
quite reliable in a healthy individual, fall down in the
critically ill patient. It is precisely in such patients
that these indicators may mislead. Skin temperature is affected
by sepsis, cardiac surgery involving systemic cooling, peripheral
vascular disease and many other factors. The kidneys themselves
may be diseased or malfunctioning, so that they cannot give
the desired information, and assumptions about SVR remaining
constant simply cannot be made in the critically ill.
2. Methods
2.1. Measuring the Cardiac Output
One might imagine that working out the
output of a pump (the heart) should be a relatively easy
task in our highly sophisticated technological age. It is
not. The simplest, most direct way would be to cut the aorta
and measure directly the output of the left ventricle. This
method may have limited use in the experimental laboratory,
but for obvious reasons is not clinically sustainable. We
are therefore forced to develop a subtler approach.
The ideal method of measuring CO should
satisfy three criteria:
- it should have accuracy, including
reliability and reproducibility
- it should be continuous, and therefore
respond rapidly to changes in CO, so that an equally rapid
therapeutic reaction can be implemented
- it should be non-invasive, or involve
as little invasion as possible
There have been many attempts at achieving
the holy grail of a non-invasive, accurate and continuous
method of measuring CO. So far, none has succeeded totally.
The remainder of this article will address the main methods
developed so far, their strengths and their weaknesses.
2.2. Fick
The Fick principle is delightful in its
simplicity and to many it states the obvious: If an organ
consumes a substance that reaches it via the blood supply,
then the amount of that substance it consumes is equal to
the blood flow to that organ multiplied by the difference
in the concentration of that substance upstream and downstream
of that organ. Any substance consumed by the body may be
used for this calculation, but the fact that the body consumes
oxygen can be put to practical use by adapting Fick to the
following:
| | | [oxygen consumption] |
| CO | = |
|
| | | [arterial oxygen content venous oxygen content] |
The three factors in this equation can
all be measured with more or less readily available tests
as follows:
Oxygen consumption can be measured by
analysing the oxygen content of inspired and expired gases
and minute ventilation volumes. Arterial oxygen content
is the total oxygen carried by haemoglobin or Hb (Hb concentration
x arterial oxygen saturation (SaO2) x constant) plus the
total dissolved oxygen in the blood (arterial partial pressure
of oxygen x constant). Venous oxygen content is calculated
in a similar way. It can be immediately seen that haemoglobin
concentration, oxygen saturations and partial pressures
are all readily available to the physician. Inspired and
expired gas analysis is less readily available, but can
be obtained, at least in a sophisticated respiratory function
laboratory.
Another approach would be to regard the
lungs, which receive the full cardiac output, as consumers
of carbon dioxide (CO2) and to reverse the formula accordingly:
| | | [CO2 excretion] |
| CO | = |
|
| | | [venous CO2 content arterial CO2 content] |
The great advantage of using Fick is its
accuracy, except in patients where an important part of
oxygen consumption occurs in the lungs, such as severe
inflammatory lung disease, where lung oxygen use may increase
from around 2% to perhaps 10 or 15% of total consumption.
The limitations on the Fick method are in the multiple measurements
required, the sophistication of the instruments needed for
blood gas analysis and in the fact that continuous measurement
is impossible without the development of reliable continuous
monitors of haemoglobin concentration, arterial and venous
oxygen saturation and partial pressures as well as continuous
monitors of blood gas delivery and composition. It can only
be used in a ventilated patient or in a patient connected
to oxygen delivery by a completely occlusive mask. In other
words, Fick is not practical for routine clinical monitoring.
2.3. Indicator dilution
It stands to reason that if one may measure
the volume (V) of a body of liquid by diluting a known quantity
of indicator (i) in the liquid and measuring the final concentration
([i]). The volume is then:
V = i / [i]
If the volume in question is the CO, then
an indicator injected intravenously can have its concentration
measured arterially (after full mixing is assumed to have
taken place). Thus:
CO= i / ò
[i] x time
where i is the amount of indicator used
and ò [i] is the integral of the downstream (arterial) concentration of
the indicator.
The advantage of this technique is its
relative simplicity and quite good accuracy. The disadvantages
arise from the necessity of injecting an indicator (typically
a coloured dye) and of multiple withdrawals of arterial
blood samples to calculate [i] over time by photodensimetric
analysis. It is clearly not suitable for continuous monitoring,
although there is currently work in progress on indicators
capable of being measured continuously through an arterial
monitoring line.
2.4. Thermodilution
This is an ingenious adaptation of dye
dilution by using heat (or cold) as the indicator, and is
currently the gold standard in clinical cardiac output
monitoring. This possibility, first suggested by Fegler
in 1954, gained widespread use after the introduction in
the early 1970s of the Swan-Ganz pulmonary artery flotation
balloon catheter. This device inserted through a large vein
into the right atrium, then carried by the blood flow on
its own flotation balloon so that its tip lies in a proximal
pulmonary artery. Using cold as an indicator, a known volume
of fluid (the injectate) at a known, cold temperature is
introduced into the right atrium through one of the many
lumina of the catheter. The tip of the device houses a thermistor
that measures the slight drop in temperature over time as
the cooled blood flows past it. Assuming perfect mixing
of the injectate with the flowing blood between injection
upstream and temperature measurement downstream, the CO
can be calculated according to a modification of the indicator
dilution formula:
| | | [injectate volume (Tblood -Tinjectate) x constant] |
| CO | = |
|
| | | òTdt |
where T is temperature, the constant adjusts
for computation and specific heat capacity of blood and
injectate (usually 5% dextrose) and òTdt
is the integral of the recorded blood temperature difference
over time. In practice, the clinician or nurse carries out
the injection, and a computer programme performs the necessary
calculations.
The method has many advantages over Fick
and indicator dilution. Although the insertion of a pulmonary
artery catheter is quite invasive, it is useful in other
respects: the measurement of right-sided cardiac pressures
as well as the capillary wedge pressure (indicating left
ventricular filling pressure) are provided as a bonus, thus
yielding a huge amount of information about cardiac haemodynamics
and justifying the invasive nature of the procedure. It
has been shown to be reasonably accurate in the clinical
setting and has performed well in comparison with Fick and
dye dilution in trials. Its accuracy, however, may be affected
by several factors, such as variability in the volume and
temperature of the injectate affecting the signal-to-noise
ratio and the differences between observer technique and
speed of injection. In addition, any pulmonary or tricuspid
valve regurgitation and incomplete mixing of the injectate
with the blood in the right ventricle will affect readings
substantially. Common practice is to carry out three to
five injections, discard unbelievable readings and average
the rest. This leads to another disadvantage of the procedure:
5 injections of 10 mls of dextrose every hour is more than
a litre of fluid per day, a volume and glucose load that
some patients cannot tolerate, especially if other therapies
are being administered intravenously. There is also a theoretical
risk of introducing infection with every injection. The
final disadvantage is that the method is intermittent and
requires positive action by the clinician to produce a measurement.
It cannot, therefore, act as an early warning system of
deteriorating cardiac function.
2.5. Continuous thermodilution
In this relatively recent development,
the thermal load is given not as a cold injectate but as
an electrically produced heat bolus through a filament incorporated
into a pulmonary artery catheter. This allows rapid, frequent
and automatic measurements without clinician intervention.
The technique is successful but has some limitations, partly
due to the smaller temperature differences that can be safely
produced within the pulmonary artery by heating a filament
in the right atrium. This reduces the signal-to-noise ratio,
and requires the use of quite elaborate neural networks
to produce information that can be reliably translated into
CO measurements. In practice, the system takes a few minutes
to stabilise and after that time, produces readings at frequent
predetermined intervals, as often as every 30 seconds, but
these readings are in fact averages of cumulative readings
obtained during the preceding few minutes. The system can
be used satisfactorily in the critically ill [Schiller,
1990]. Its main limitation is that it is not truly continuous
and it is less accurate and reliable if the patient is pyrexial.
The level of invasiveness of the procedure is not different
from that of the intermittent thermodilution method.
2.6. Bioimpedance
The volume of blood ejected every heartbeat
is called the stroke volume. Multiplying this by the pulse
rate gives the cardiac output. The pulse rate is easily
available, and bioimpedance techniques are directed towards
measuring the stroke volume. The principles of this measurement
is that the electrical impedance to a current across any
conductor, say the thorax or the body itself, is equal to
the voltage divided by the current. The impedance to flow
can be divided into two components: that provided by fixed
tissues and body cavity content and that provided by mobile
contents, the most important of which is the cardiac stroke
volume. To put it simply, the variation in impedance across
the chest should in principle reflect the volume of blood
ejected with every heart beat, and therefore give an indirect
measurement of stroke volume.
A number of bioimpedance CO monitors have
been developed. Appropriately placed electrodes deliver
a certain current across the chest and accurately measure
the changes in the voltage across the cardiac cycle. These
changes are then adjusted using normograms of thoracic volume
and proportion of electrically active tissue based on height,
weight and sex of the patient to yield an estimate of the
stroke volume and thus of the cardiac output.
The great attraction of bioimpedance technology
is that it is non-invasive and can theoretically be continuous
on a beat-by-beat basis. Unfortunately, correlation with
other methods has not been consistently satisfactory in
clinical trials and the method is subject to further errors
in patients with dysrhythmia or pulmonary or pleural conditions
that affect thoracic water volume, such as oedema or effusions,
conditions seen all too frequently in the critically ill.
2.7. Doppler
Doppler echocardiography provides an alternative
noninvasive method of measuring total forward blood flow
and hence cardiac output. This method relies on combining
imaging data from 2-dimensional echocardiography and flow
data from spectral Doppler echocardiography. Spectral Doppler
allows instantaneous blood flow velocity to be measured,
which is expressed in units of distance per time (m/sec).
In order to quantitate this as a volume per time i.e. L/min,
the velocity must be multiplied by the cross-sectional area
of the orifice or tube through which the blood is flowing
(2)
Cardiac Output (L/min)= A
x V
where
A = cross-sectional area of orifice or tube (cm2)
V = mean blood flow velocity (cm/sec)
Here we are considering velocity averaged
over the cardiac cycle. However spectral Doppler measures
instantaneous velocity. Therefore to derive mean velocity
over the cardiac cycle, we measure the area under the velocity
curve during one heartbeat, the velocity time integral (VTI),
and multiply by heart rate (3)
Cardiac Output (L/min)=A
x VTI x HR
Echocardiography is commonly performed
by either the transthoracic or transoesophageal approach.
By Doppler echocardiography flow can be measured in numerous
locations in the heart, through the heart valves, and the
great vessels.
There are several other Doppler techniques
now currently available although not in widespread clinical
use. These include a new pulmonary artery catheter that
incorporates an ultrasonic transducer that maintains contact
with the pulmonary artery wall. Using the Doppler principle,
instantaneous stoke volume is obtained from the mean velocity
of blood flow in the main pulmonary artery [Segal et al.,
1990; Abrams et al., 1989]. Ultrasonic transducers have
also been bonded to endotracheal tubes, which by contact
with the wall of the trachea are in close proximity to the
ascending aorta where blood flow is measured similarly to
transoesophageal Doppler [Rodig et al., 1999].
2.8. Pulse Contour
A new system of pulse contour analysis
is now also available, which is less invasive than continuous
CO monitoring via a pulmonary artery catheters, and may
cost less. The notion that SV (stoke volume) can be quantified
from pulse pressure goes back to observations by Erlanger
and Hooker in 1904. This technique requires the insertion
of an arterial thermodilution catheter via the femoral artery
into the aorta for clinical monitoring of the arterial pressure,
continuous CO measurements derived from the arterial pressure
and intermittent arterial thermodilution CO measurements.
[Boldt et al., 1994]
Pulse contour analysis involves calculating
the stroke volume from the contour of the arterial waveform.
The area under the systolic portion of the arterial pulse
wave is measured from the end of diastole to the end of
the ejection phase, together with an individual calibration
factor to account for individual impedance. To calibrate
this system, individual arterial input impedance to arterial
pressure is calculated from the area under the systolic
potion of the curve arterial pulse wave, and the arterial
thermodilution CO. A disadvantage of this technique is that
assumptions are made concerning the distensibility of the
systemic vascular bed. [Segal et al., 1989]
2.9. Heat Transfer
This new technique, towards which one
of the authors (SAMN) is biased, is based on the simple
notion that any heat dissipated by the flow of blood is
proportional to that flow. The method uses a pulmonary artery
flotation catheter (TruCCOM, Aortech, Scotland) which is
inserted and advanced into the main pulmonary artery in
the same way as a thermodilution catheter. The catheter
differs from the standard thermodilution catheter in two
ways: firstly, it has two thermistors, one at the tip in
the usual position and another proximal (in the right atrium)
to the first; secondly, the first thermistor is surrounded
by an electrical heating coil. A current is applied to the
heating coil to warm the first thermistor to a set temperature
slightly higher (1-2 degrees) than that of the blood, as
measured by the second thermistor. The amount of heat thus
generated is negligible, and does not appreciably alter
the temperature of the blood, but the power needed to maintain
the temperature difference is proportional to the blood
flow that is constantly cooling the coil, in other words
, the CO.
This type of heat transfer technology
has been used in the past to measure aircraft speed in what
is called hot-wire anemometry. It can therefore be argued
that the method measures velocity more than it measures
flow. This argument would be correct if flow through the
pulmonary artery was laminar, and the coil was only exposed
to a thin stratum of blood during the cardiac cycle. However,
pulmonary artery flow is anything but laminar: it is pulsatile
with both to-and-from motions and eddy currents, and it
is made turbulent by the opening and closing actions of
the pulmonary valve. This means that the coil is exposed
to more than a thin stratum of laminar-flowing blood, and
the method reflects flow better than it reflects velocity.
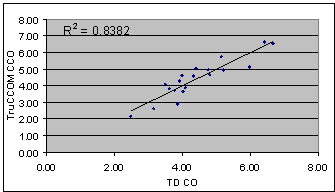
Figure 1. Correlation between simultaneous
readings of cardiac output using continuous (TruCCOM) and
standard thermodilution (TD). Flows in litre per minute.
The heat transfer technique has been tested
in vitro and in experimental animals and has performed well.
Early clinical trials suggest that it correlates well with
the current clinical gold standard of thermodilution (Fig.
1). More importantly, it is probably the only method available
that is capable of responding to cardiac output changes
within seconds, thus providing clinicians with valuable
early warning in deteriorating cardiac output as well as
a rapid feedback on the results of their therapeutic intervention
(Fig. 2). It is probably the only method which provides
truly continuous measurement of CO, and the rapidity of
its response has opened possibilities in cardiac output
monitoring in situations where such measurement has not
been practicable by other techniques because of their inherent
slowness, such as during percutaneous coronary angioplasty
and off-pump coronary artery surgery
The remaining disadvantage
is, of course, that the procedure is still an invasive one,
requiring the insertion of a pulmonary artery catheter.
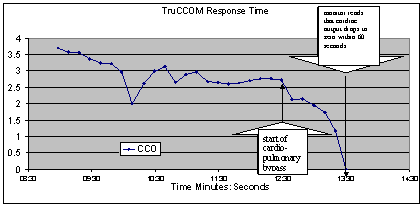
Figure 2. Response of heat transfer cardiac output monitor to the start of cardiopulmonary
bypass in a cardiac surgical patient (CCO: continuous cardiac
output, litres per minute).
3. Conclusion
Despite the recent controversy surrounding
the necessity and clinical usefulness of cardiac output
monitoring in the critically ill, most clinicians agree
on the need for such monitoring in order to guide therapy.
It would be true to say that the perfect, 100% safe, non-invasive,
continuous and fully accurate cardiac output monitor has
not yet been invented. Many strides have been made towards
this goal, and the multiplicity, substantial successes and
sheer ingeniousness of the recently developed and currently
available methods augur well for the future.
References
Abrams JH, Weber RE, Holman KD. Transtracheal
Doppler: A new procedure for continous cardiac output measurement.
Anesthesiology, 70:134, 1989.
Boldt J, Menges T, Wollbruck M, Hammermann
H, Hempelmann G. Is continuous cardiac output measuerement
using thermodilution reliable in the critically ill patient?
Crit Care Med, 22: 1913-1918, 1994.
Kaplan JA. Cardiac Anesthesia, 3rd Ed.
WB Saunders Company, 1993.
Rodig G, Prasser C, Keyl C, Liebold A
and Hobbhahn J. Continuous cardiac output measurement: pulse
contour analysis vs thermodilution technique in cardiac
surgical patients. British Journal of Anaesthesia,
82(4): 525-530, 1999.
Schiller NB. Cardiology Clinics Doppler
Echocardiology. WB Saunders Company, 1990.
Segal J, Pearl RG, Ford AJ et al: Instantaneous
and continuous cardiac output obtained with a Doppler pulmonary
artery catheter. J Am Coll Cardiol, 13:1382, 1989.
Segal J, Nassi M, Ford AJ, SchuenemeyerTD.
Instantaneous and continuous cardiac output in humans obtained
with a Doppler pulmonary artery catheter. J Am Coll Cardiol,
16:1398,1990.

 Home
Current Issue
Table of Contents
Home
Current Issue
Table of Contents