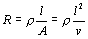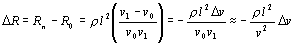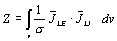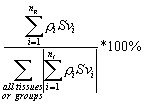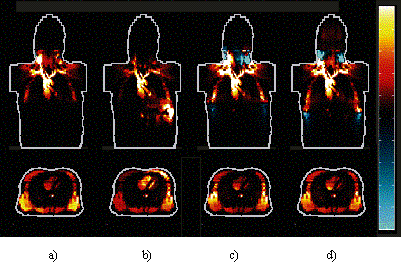Pasi K. Kauppinen(a),
Jari Hyttinen(a), Tiit Kööbi(b),
Seppo Kaukinen(c),
Jaakko Malmivuo(a)
Correspondence: PK Kauppinen,
Ragnar Granit Institute, Tampere University of Technology,
P.O. Box 692, FIN-33101 Tampere, Finland.
E-mail:
pasi.kauppinen@tut.fi,
phone +358 3 247 4012, fax +358 3 247 4013
1. Introduction
An ideal method of assessing information on the cardiovascular
system should be noninvasive, simple, atraumatic, inexpensive,
reliable and also applicable in long-term surveillance outside
the cardiac monitoring laboratory. Conventional impedance
cardiography (ICG) techniques provide a single impedance
tracing, from which parameters related to the pump function
of the heart such as cardiac output (CO) are estimated.
Most of the properties of ICG render it superior to other
methods, the prominent exception being its limited reliability,
which has hampered its acceptance as a clinical method.
When the notion of impedance is transferred to the biological
tissues in the human body and to applications seeking to
quantify physiological function from measured data, the
investigator moves into an area complex and incompletely
understood.
1.1. Conventional ICG
Measurement of thoracic electrical impedance has been practiced
since the 1930s [Nyboer, 1970]. The first practical method
for determination of cardiac function in a clinical setting
was introduced by Kubicek et al. in the 1960s, together
with the original CO formula based on elementary physics
[Kubicek, 1989; Kubicek et al., 1966; Patterson et al.,
1964]. Several variations in electrode configurations and
CO equations have been presented over the years to improve
the method [Bernstein, 1986; Penney et al., 1985; Woltjer
et al., 1995].
Basic Principle
The impedance ( Z ) is
a function of the cross-sectional area ( A ) and
length ( l ) between
the voltage pick-up electrodes applied on the volume conductor.
In terms of segmental volume (v), ( Z ) or
alternatively resistance R is given
by
| |
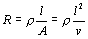 |
(1) |
where l is the
voltage pick-up electrode separation.
Nyboer [Mohapatra, 1981; Nyboer, 1970] assumed that the
pulsatile flow of blood could be modeled as the resistance
of the blood pulse, R b, in parallel
with the steady basal value of resistance, R 0, of
the surrounding tissue, forming a total resistance R n
The resistance change ΔR due
to the addition of blood volume Δv is derived
from Eq. 1 as follows:
| |
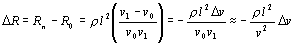 |
(2) |
where v0 is
the original volume of the object and v1 the
volume after the addition of blood, which for small changes
in v is v0 ≈ v1. Substituting
Eq. 1 and rearranging, Nyboers formula is obtained
| |
 |
(3) |
This equation describes the relationship between the volume
of a blood pulse and the related resistance change. The
model constitutes an oversimplified view of any physiological
system.

Figure 1. Various electrode configurations for
ICG reported in the literature. a) the conventional band,
b) four-spot, c) eight-spot and d) nine-spot electrode configurations.
Locations of black spots are used for current injection
and white spots for voltage detection in actual measurements.
Various ICG Methods
Since the functioning of the heart is obviously the
generator of measurable cyclic impedance variation, many
investigators have sought to evolve methods for CO derived
from this periodic signal applying various modifications
to the Eqs. 1 3 or electrode configurations. Fig. 1 shows
several configurations reported in the literature. The well-established
band electrode method by Kubicek has been replaced by various
spot electrode configurations in the effort to improve patient
comfort and the practicality of the method without significantly
changing the signal content.
1.2. Field Theoretical Analyses of ICG
Although much work has been done to date, the development
of ICG is still principally based on the same ideology introduced
decades ago, relying strongly on an empirical approach.
Use of over-simplified models has limited and confined improvement
in the technique because of the gap between the model and
the anatomy and physiology of the system investigated.
Computer models designed to calculate the current flow
in the thorax have been used more recently in examining
the ICG measurement configurations, producing supporting
data for the anticipated conception of the complexity of
the signal origin [Kim et al.,1988; Wang and Patterson,
1995; Wtorek and Polinski, 1995].
A theoretical foundation exists for analysis of the measurement
sensitivity distribution of ICG based on the lead field
theory introduced in the 1950s for bio-electric (i.e. ECG)
analysis and later in the 1970s [Geselowitz, 1971] for bio-impedance
(BI) measurements. BI measurement sensitivity distribution
reflects how conductivity changes throughout the volume
affect the measured data. The application of lead field
theory in ICG has not evolved to its potential; only initial
studies with analytical models have hitherto been conducted
to estimate the measurement sensitivity distribution in
cylindrically shaped objects with uniform conductivity [Penney
et al., 1979].
1.3. Objectives of this Study
The purpose here was to utilize the lead field theoretical
approach in investigating the information content of existing
ICG measurement configurations, and in examining possibilities
of recording ICG within the clinical setting of the 12-lead
ECG based on computerized application of the lead field
theory in realistically shaped volume conductors.
2. Material and Methods
2.1. ICG Sensitivity Distribution
The sensitivity distribution of a BI measurement gives
a relation between the impedance (and change in it) caused
by a given conductivity distribution (and its change). It
describes how effectively each region is contributing to
the measured Z. If conductivity
change is not involved, the measured impedance Z is obtained
with
| |
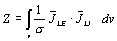 |
(4) |
where JLE
and JLI, obtained
with reciprocal energization, are the current density fields
(i.e. impedance measurement lead fields) associated
with the current injection and voltage measurement leads
[Kauppinen, 1999; Malmivuo and Plonsey, 1995]. This equation
gives the contributions from each region to the total impedance,
and the dot product of the two fields expresses the sensitivity
of the measurement to conductivity changes throughout the
volume conductor.
The impact of a certain conductivity variation in different
regions depends on the sensitivity field. As the scalar
field may possess positive and negative values depending
on the orientation of the two lead fields, the measured
impedance may either increase, decrease or be entirely unaffected
in consequence of a conductivity change in a particular
region.
The relative magnitude of the sensitivity field in a tissue
type (or a group of tissues considered as one target volume)
gives a measure of how conductivity variation in that tissue
will affect the detected DZ. The overall sensitivity of
a tissue type is obtained by integrating the sensitivity
values of the tissue over the volume it occupies. This sensitivity
value can then be compared with the absolute total sensitivity
of the model as given by
| |
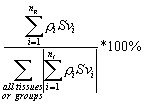 |
(5) |
where ng is the number of elements in
the target volume and nt the number of
tissue elements of a certain type. The denominator is the
sum of the absolute partial contributions from all tissues
(or tissue groups), and the numerator is the contribution
of the target tissue.
2.2. Volume Condutor Computer Models
Utilizing Eq. 4 with finite difference method (FDM) computer
modeling, information as to the respective capacity of different
ICG measurements to detect conductivity and its changes
in the thorax can be estimated. Methods to construct and
solve accurate volume conductor computer models based on
the FDM have been previously developed and validated [Kauppinen
et al., 1999].
Three different anatomy models were employed in the study:
Visible Human Man model.
A particularly accurate
source of anatomical data, the U.S. National Library of
Medicines Visible Human Man (VHM) [Ackerman et al., 1991;
Spitzer et al., 1996] was employed as basis for detailed
FDM modeling. The original cryosection images are 2048 by
1216 pixels in 24-bit color, resulting in about 14 gigabytes
of data in size. A total of 118 cryosection images from
the top of the head to the pelvis were segmented using a
volume segmentation method which directly provides volume
elements of anatomy data for FDM mesh generation [Heinonen
et al., 1998]. For data storage and image analysis the accuracy
of the images was reduced to 250x250 pixels using an 8-bit
gray scale colormap. The resolution was from 0.044 to 5.7
cm3.
Dynamic Model - Diastolic & Systolic models.
The ECG triggered end-systolic and end-diastolic MR image
data sets used by Wang and Patterson [Wang and Patterson,
1995] were segmented. A two-phase thorax model (i.e. two
models of the same person at different moments of the cardiac
cycle) was constructed from these data producing end diastole
model (EDM) and end systole model (ESM). The number of voxels
was equal in both models, as the segmented outermost layer
from the first data set was used as base for segmentation
of the other set. Both models consisted of 70 slices and
30 tissue types. The resolution of FDM elements in the ECG-triggered
models varied from 0.10 to 5.8 cm3 resulting
to 121431 elements.
2.3. Analysis of Conventional ICG Electrode Configutations
Contributions to the sensitivity distribution were assessed
with the VHM thorax model for four ICG electrode configurations
utilizing conventional band electrodes or modifications
replacing the bands with spot electrodes (see Fig. 1):
a) Original configuration by Kubicek et al. using four
band electrodes [Kubicek et al., 1966]
b) Configuration by Penney et al. using four spot electrodes
[Penney et al., 1985]
c) Configuration by Bernstein using eight spot electrodes
[Bernstein, 1986]
d) Configuration proposed by Woltjer et al. using nine
spot electrodes [Woltjer et al., 1996].
Simulations were conducted to obtain the basal impedance,
Z0, lead fields in the thorax generated by the
current and the measurement leads
JLE
and JLI,
and the resulting measurement sensitivity distribution according
to the Eqs. 4 and 5.
2.4. Derivation and Analysis of 12-lead ICG Measurement
Configurations
The prospects of recording multiple ICG waveforms with
more selective sensitivity to particular regions of the
thorax were investigated employing the 12-lead electrode
system. The lead field concept can be applied, facilitating
synthesis of leads with desired properties, e.g., more specific
leads.
The nine electrode locations of the 12-lead ECG electrode
system were used separately to calculate a basic set of
lead fields for each model, the VHM and two-phase models.
A computer algorithm was developed to make combinations
with the 12-lead electrode system using at maximum four
electrodes at a time for either lead field in Eq. 4. Deriving
ICG measurement combinations with the pre-calculated lead
fields is a non-iterative calculation, since the system
is assumed to be linear. E.g. a lead field between the chest
leads V1 and V6 may be obtained by subtracting V1LL
from V6LL. A total of 65476 impedance measurement
configurations utilizing the 12-lead electrode locations
was thus derived.
A database was computed for each model and measurement
configuration containing the information on the formation
of Z0 and proportional contributions according
to Eq. 5. This was done for each classified tissue and for
a number of different tissue groups reflecting functional
structures of the cardiovascular system. Further, the same
calculations were applied to the data produced by subtracting
the sensitivity and Z0 values simulated by the
ECG-triggered models EDM and ESM.
2.5. Experimental Clinical Measurements
An experimental pilot study was conducted to evaluate the
impedance waveforms detected by the 12-Lead configurations
selected based on the simulations. The study involved 12
healthy volunteers and a group of 9 patients with valvular
heart disease (3 mitral, 6 aortic).
The measurements were performed by CircMonä B202 (JR medical
Ltd, Tallinn, Estonia), which includes an impedance channel
delivering 0.7 mA at 30 kHz in combination with a devised
front-end apparatus [Kauppinen et al., 1999], that enables
to record multiple 12-lead ICG signals with digitally controlled
electrode configurations.
2.6. Analysis and Comparison of Clinical and Simulated
Data of 12-Lead ICG
The theoretical data from the models were compared between
each model and the data from the test measurements. The
clinical data was compared between the two study groups
consisting of healthy volunteers and valvular patients.
Triggering to the R-peak of ECG, ensemble averaged measured
DZ signals were calculated for each configuration and subject.
Several parameters were derived from the data measured from
the volunteers: minimum and maximum Z0, DZmax
indicating the maximum impedance deflection of averaged
DZ, DZvent the amplitude of ventilation effect
during the recording period of each configuration, ratio
of DZmax/DZvent, and the mean absolute
percentage error (MAPE) between the individual average DZ
and the average DZ calculated from all the volunteers. MAPE
describes the difference between an individual DZ and the
average DZ obtained from the study group with particular
configuration. The parameters were computed as average values
from each subject for each configuration. Correlation matrix
was calculated between the derived parameters and the simulated
sensitivity distributions. In addition, the inter-group
(volunteers versus patients) differences (MAPEs) were calculated
for the averaged DZs.
3. Results
3.1. Simulated Sensitivity Distributions
Conventional ICG electrode configurations
The conventional band electrode configuration and alternative
configurations suggested to replace the bands were shown
not to be specifically sensitive in measuring conductivity
changes in regions generally considered important in measuring
SV or CO, namely the heart, lungs or aorta and other large
vascular trees. More than half of the measurement sensitivity
in each case studied was concentrated in the skeletal muscle,
while less than 5 % was in the heart region and the large
vessels. Furthermore, the results showed heterogeneous current
field flow in the thorax, and modifying the electrode configuration
resulted in different sensitivity distributions which must
have an influence on the composition of measured signals.
This is further illustrated in the frontal and transversal
sensitivity images shown in Fig. 2. Sensitivity at the mid-frontal
plane was most uniform with the band setting and least uniform
with the eight-spot setting. Using the band electrodes the
transversal sensitivity image showed a significant source
of signal from the posterior side of the thorax. The four-spot
configuration caused a more concentrated current flow in
the left side of the thorax, which increased the sensitivity
on that side.
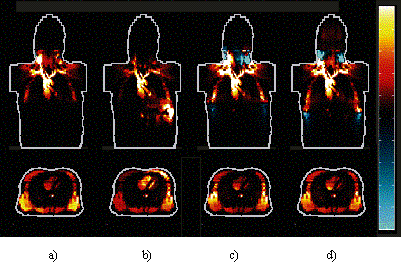
Figure 2. Mid-frontal and transversal views of
sensitivity field distributions. a) conventional band, b)
four-spot, c) eight-spot and d) nine-spot configurations.
Zero sensitivity is indicated with black colour, positive
sensitivities are visualized with hot colourmap and negative
with cool colourmap.
12-Lead ICG
Simulations revealed that as compared to conventional
ICG, clearly enhanced sensitivities can be obtained in various
regions of the cardiovascular system by appropriately selecting
the measurement configurations used for ICG. Figure 3 summarises
the simulated measurement sensitivities of the configurations
discussed above. Values are indicated for each tissue type
in addition to three tissue groups consisting of pulmonary
circulation, systemic circulation and all the blood masses
and the heart muscle. For the tissues of the cardiovascular
structures, a maximum of 75 % proportional sensitivity was
attained. The agreement between the different models was
notable and the values in Fig. 3 are averages as calculated
from the three models.
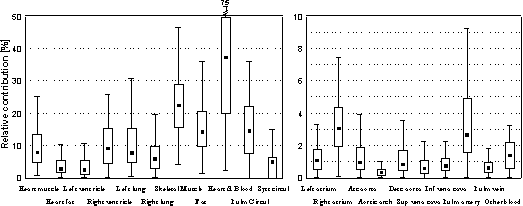
Figure 3. Sensitivities in different tissues
for the configurations selected for clinical experiments.
Values are calculated as averages from the three different
models. The median of the values is represented by the smallest
box in the plot, the spread (variability) by the quartiles
(the 25th and 75th percentiles) and the minimum and maximum
values of the sensitivity among the configurations.
3.2. Correlation Data
For the Z0 the simulated values showed statistically
significant correlation between the models (p<0.008).
The calculated correlation matrix showed a large number
of statistically significant relationships between the simulated
data and the parameters derived from the measurements. The
strongest value for correlation was 0.45 as calculated between
the positive area after the R-peak and the sensitivity proportion
in right ventricle in ESM (p<0.000) and also for the
MAPE after the R-peak and the Z0 change between
the ECG triggered models (p<0.000). DZmax
did not show correlation to Z0 simulated with
any of the models, but significant correlation was found
to the DZ calculated by subtracting values of Z0
calculated with the EDM and ESM (r=0.30, p<0.000). Proportional
sensitivity change as calculated with the EDM and ESM correlated
positively with DZmax when investigating the
heart region (r=0.31, p<0.000), while negative correlation
was found for the sensitivity in the lungs.
3.3. Inter-Individual Variations within Volunteers
Generally, increasing MAPEs correlated positively with
the Z0 difference between the triggered models
(r=0.24
0.44, p<0.000), the sensitivity change in the
fat and skeletal muscle (r=0.24
0.43, p<0.000) and also
the heart region r=0.29
0.37, p<0.000). Negative correlations
(i.e. smaller MAPE with higher sensitivity) were evident
for lung configurations
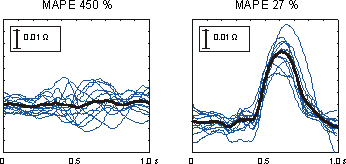
Figure 4. DZ waveforms obtained with two different
configurations exhibiting the smallest and largest variations
(MAPE) between the volunteers. Thin lines show individual
averaged DZ, thicker tracing the average from all the volunteers.
with all of the three models (r=-0.23
-0.32,
p<0.000), but not for the sensitivity change obtained
by subtracting the triggered model values, and for the tissue
groups consisting of either pulmonary or systemic circulation.
Fig. 4 shows averaged DZs from the volunteers for the configurations
producing the smallest (27%) and the largest (450%) inter-individual
MAPEs for the period of the whole cardiac cycle.
3.4. Differences Between Volunteers and Patients
The inter-group MAPEs between the volunteers and the valvular
patients showed the highest positive correlation to the
sensitivity in VHM heart muscle (r=0.36, p<0.000). MAPE
before the R-peak correlated positively to the sensitivity
change between the triggered models in the atria (r=0.38,
p<0.000), and after the R-peak to the heart muscle and
left ventricle (r=0.20, p<0.003). The largest negative
correlation was found for the sensitivity in the lungs in
the triggered models (r=-0.25, p<0.000). Example averaged
DZs recorded from both of the study populations are shown
in Fig. 5.
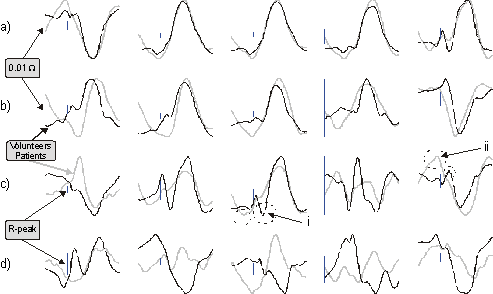
Figure 5. Examples of the 12-lead based ICG recordings
shown as average signals from the study groups (volunteers
and valvular patients). a) Tracings with small inter-group
variation, b) changes between the groups in the time instant
of the maximum impedance deflection, c) characteristic signals
with notable peaks or deflections missing between the groups,
d) large inter-group MAPEs. For explanation of tracings
marked i and ii, please refer to text.
Certain configurations recorded similar waveforms
between the study groups (a), several produced similar shapes
in the impedance deflection but shifting the time instant
of the peak change (b). The maximum impedance deflection
was more often delayed than advanced with the patients when
compared to the healthy subjects although the patients heart
rate was higher (average HR: volunteers 62, patients 71).
Fig. 5 c) shows configurations with additional or missing
deflections between the study groups, and d) data with larger
deviations between the groups. Investigating DZs from individuals
reveals a tendency shown in the averaged signals, although
some configurations seem to detect regional information
and thus producing more diversified impedance curves. As
an example, in (c) the distinct peak marked i occurs in
the data recorded from 11/12 volunteers, but not in any
patient. The peak marked ii, on the other hand, is elevated
in 6/9 patients and in only one volunteer.
4. Discussion
Computer Models.
The anatomical differences between
the VHM and the two-phase models were considerable; for
instance, the total volumes were 47 and 21 l for the VHM
and the two-phase models, respectively. In spite of this,
the most selective configurations for certain anatomical
region or tissue were identical independent of the model
applied. This result was unexpected, since to achieve high
sensitivity in a certain region and low in others requires
in principle a measuring configuration where lead fields
of current and voltage electrodes are practically perpendicular
to each other. Slight deviations in model geometry or electrode
locations could then markedly modify the partial sensitivity
values.
Conventional ICGs.
Simulation results emphasized
the multiregional sampling sensitivity of the studied ICG
configurations. For the conventional ICGs, only an approximately
5 % contribution from all blood masses and cardiac tissue
was detected. This can be taken to imply that the valuable
information, i.e. the information needed to determine the
CO or other desired parameters, overlaps with a wide range
of other information unretrievable from DZ. Although useful
information has been obtained from the ICG waveform, it
originates from a multiplicity of sources with unpredictably
varying contributions depending on the characteristics and
hemodynamic condition of the subject. Thus, particular caution
is called for when applying ICG to clinical work. It is
unlikely that a universally ideal electrode configuration
providing accurate measurements exists for ICG. If the number
of unpredictable factors contributing to or modifying the
ICG waveform are many, at least as many specific measurements
should be taken as there are contributing factors to establish
the state of the system.
12-Lead ICG.
Numerical modeling with the lead field
theoretical approach made possible detailed analysis of
a large number of configurations. Increasing the contribution
from a limited region may improve the physiological relevance
of recorded data, which was achieved in theory with the
regional multi-electrode measuring configurations. According
to simulations, for the tissues of the cardiovascular structures,
a maximum of 75 % proportional sensitivity was attained.
For the aortas and vena cavas the values were relatively
small, since the electrode locations of the 12-lead system
are not favorable for vertical measurements. Although highly
elevated sensitivities were obtained, it was not possible
to achieve fully selective measurements for any of the tissues.
Moreover, even with these enhanced measuring configurations,
everything still affects everything; nonetheless their relative
contributions should be more favorable than in conventional
methods to produce regional information.
Clinical Experiments.
Weak, but statistically significant
relationships between the simulated data and the parameters
derived from the measurements were attained indicating the
general ability of the modeling approach in developing and
understanding the properties of various electrode configurations
applied in ICG. Recorded 12-lead signals had characteristic
landmarks not coincing with those of conventional ICG, indicating
varied information content between the configurations. Furthermore,
signals were noted showing a suggestive resemblance to invasive
data and morphological variations in disease not present
with conventional ICG. Valvular disease was detected when
investigating at least two signals simultaneously; a single
ICG signal cannot produce information for the identification
of the existence of the disease. An important limitation
in the clinical measurements is the instrumentation restricting
the analysis of collected signals since only one channel
can be recorded at a time.
Future Research.
On the theoretical side, extending
the two-phase to a multi-phase model one could simulate
the ΔZ waveform
and investigate geometrical and conductivity changes separately
and not only assess the sensitivity distribution at one
time instant. Comparisons with clinical data would facilitate
the development of models and provide a guide in selecting
valid tissue conductivities. Further research is clearly
needed to determine the precise effects of 1) inter-individual
variations, 2) postural changes and 3) breathing on a) sensitivity
distributions and b) measured data. Future studies should
also take account of the effects of slight positional changes
of electrodes. To collect a wide range of clinical data
also from patients during invasive measurements, the system
should be implemented in a multi-channel form allowing parallel
recording of all independent impedance signals. A
multi-channel ICG instrumentation would be practical in
prospective 12-lead measurements reducing the overall recording
time and allowing analyses of signals recorded simultaneously.
A large diverse group of patients in each disease category
should be investigated pre- and postoperatively prior to
drawing any definite conclusions about the 12-lead ICG measurements.
5. Conclusions
Estimation of CO from a non-invasively measured impedance
signal is an ill-posed inverse problem with no unique solution,
since DZ is always a combined presentation of multiple sources.
An ICG method could be made more reliable by sampling the
target region with several electrode settings giving emphasis
to the particular region. The results obtained demonstrate
the feasibility of the lead field method in developing ICG.
Recorded 12-lead ICG signals exhibited landmarks not coincing
with those of conventional ICG showing morphological variations
in disease. Configurations producing regional information
may have a range of applications apart from the CO estimation.
However, understanding the information conveyed by the 12-lead
ICG requires multi-phase modelling and measurements with
simultaneous acquisition of hemodynamic variables such as
flow and pressure in the structures of the cardiovascular
system. This calls for more advanced multi-channel instrumentation
for testing the methodology in clinical environment.
Acknowledgements
This work was supported financially by the Ragnar Granit
Foundation. The authors wish to thank Professor Robert Patterson,
University of Minnesota, for supplying the MRIs for the
two-phase model.
Ackerman MJ. The Visible Human Project. Journal of Biocommunications,
18: 14, 1991.
Bernstein DP. A new stroke volume equation for thoracic
electrical bioimpedance: theory and rationale. Critical
Care Medicine, 14: 904-909, 1986.
Geselowitz DB. An application of electrocardiographic lead
theory to impedance plethysmography. IEEE Transactions
on Biomedical Engineering, 18: 38-41, 1971.
Heinonen T, Dastidar P, Kauppinen P, Malmivuo J and Eskola
H. Semi-automatic tool for segmentation and volumetric analysis
of medical images. Medical & Biological Engineering
& Computing, 36: 291-296, 1998.
Kauppinen P. Application of lead field theory in the analysis
and development of impedance cardiography. PhD Thesis, Tampere
University of Technology, Tampere, Finland, 1999.
Kauppinen P, Hyttinen J, Laarne P and Malmivuo J. A software
implementation for detailed volume conductor modelling in
electrophysiology using finite difference method. Computer
Methods and Programs in Biomedicine, 58: 191-203, 1999.
Kauppinen PK, Hyttinen JAK, Kööbi T and Malmivuo J. Multiple
lead recordings improve accuracy of bio-impedance plethysmographic
technique. Medical Engineering & Physics, 21:
371-375, 1999.
Kim DW, Baker LE, Pearce JA and Kim WK. Origins of the
impedance change in impedance cardiography by a three-dimensional
finite element model. IEEE Transactions on Biomedical
Engineering, 35: 993-1000, 1988.
Kubicek WG. On the source of peak first time derivative
(dZ/dt) during impedance cardiography. Annals of Biomedical
Engineering, 17: 459-462, 1989.
Kubicek WG, Karnegis JN, Patterson RP, Witsoe DA and Mattson
RH. Development and evaluation of an impedance cardiac output
system. Aerospace Medicine, 37: 1208-1212, 1966.
Malmivuo J and Plonsey R. Bioelectromagnetism: Principles
and Application of Bioelectric and Biomagnetic Fields. Oxford
University Press, New York, 1995.
Mohapatra SN. Non-invasive Cardiovascular Monitoring by
Electrical Impedance Technique. Pitman, London, 1981.
Nyboer J. Electrical Impedance Plethysmography. Charles
C Thomas, Springfield, 2nd ed,1970.
Patterson RP, Kubicek WG, Kinnen E, Witsoe DA and Noren
G. Development of an electrical impedance plethysmography
system to monitor cardiac output. In proceedings of the
1st Annual Rocky Mountain Bioengineering Symposium, 1964,
56-71.
Penney BC, Narducci LM, Peura RA, Anderson FAJ and Wheeler
HB. The impedance plethysmographic sampling field in the
human calf. IEEE Transactions on Biomedical Engineering,
26: 193-198, 1979.
Penney BC, Patwardhan NA and Wheeler HB. Simplified electrode
array for impedance cardiography. Medical & Biological
Engineering & Computing, 23: 1-7, 1985.
Spitzer V, Ackerman MJ, Scherzinger AL and Whitlock D.
The visible human male: a technical report. Journal of
the American Medical Informatics Association, 3: 118-130,
1996.
Wang L and Patterson R. Multiple sources of the impedance
cardiogram based on 3-D finite difference human thorax models.
IEEE Transactions on Biomedical Engineeringg, 42:
141-148, 1995.
Woltjer HH, Arntzen BW, Bogaard HJ and de Vries PM. Optimalisation
of the spot electrode array in impedance cardiography. Medical
& Biological Engineering & Computing, 34: 84-87,
1996.
Woltjer HH, van der Meer BJ, Bogaard HJ and de Vries PM.
Comparison between spot and band electrodes and between
two equations for calculations of stroke volume by means
of impedance cardiography. Medical & Biological Engineering
& Computing, 33: 330-334, 1995.
Wtorek J and Polinski A. Examination of impedance cardiography
properties--FEM model studies. Biomedical Sciences Instrumentation,
31: 77-82, 1995.

 Home
Current Issue
Table of Contents
Home
Current Issue
Table of Contents