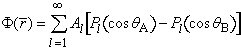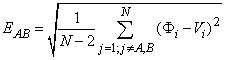Thomas C. Ferreea,b and Don M. Tuckera,b
1. Introduction
The development of high-resolution electroencephalography (EEG) involves advancements in both
device technology and analysis methods and software. Regarding devices, the efforts of our group are focused mainly
on the development of the Geodesic Sensor Net and the Net Amps amplifier system. The Geodesic Sensor Net (GSN)
is a dense sensor array of 128 or 256 electrodes with approximately equal spacing between adjacent pairs [16].
Figure 1 shows that, in addition to providing high spatial resolution, the design of this net solves a number of
practical problems. It is quick and easy to apply, does not require skin abrasion, and can be worn comfortably
for hours. In addition to ease of use in research settings, recent studies at the Sacred Heart Medical Center in
Eugene, Oregon, show that the GSN is also practical and reliable for clinical use [8].



Figure 1. Application of the 128-channel Geodesic Sensor Net.
The voltage signals are amplified by the Net Amps, which are designed specifically for use with
the GSN. While high-resolution EEG promises to enable researchers and clinicians to study patterns of brain activity
not detectable with conventional electrode arrays [14], several technological challenges remain to make maximal
use of this rich data.
Primary among these challenges is the fact that accurate three-dimensional spatial
analysis of EEG data via electric head models requires accurate representation of both head geometry and regional
head tissue conductivity [1,11,12,15]. Although early head models assumed spherical head geometry, modern boundary
element [10] and finite element methods allow the incorporation of realistic head geometry. Modulo the technical
challenges of automated tissue segmentation, structural MRI can provide the necessary anatomical information for
accurate head models. In both spherical and realistic models, the head is usually represented electrically as four
homogeneous and isotropic conductive regions. Table 1 shows the ranges of these regional tissue conductivities,
tabulated from the experimental literature [2,5,7].
Table 1. Head tissue conductivity (1/Ωm)
tabulated from the
experimental literature.
| Tissue |
Mean σ |
Stdev σ |
Min σ |
Max σ |
| Brain |
0.25 |
0.13 |
0.05 |
1.0 |
| CSF |
1.79 |
0.02 |
1.73 |
1.85 |
| Skull |
0.018 |
0.014 |
0.002 |
0.1 |
| Scalp |
0.44 |
0.2 |
0.05 |
1.0 |
With the possible exception of the cerebrospinal fluid (CSF), the variability in these data are
greater than the precision thought to be required for accurate EEG analysis [1,11,12,15]. Nevertheless, most researchers
continue to take conductivity parameters from these standard references, presumably because there is no better
method currently available.
We have therefore developed a method of in vivo regional head tissue conductivity estimation,
which can easily be applied to individual subjects [4]. In this method, suggested previously by Eriksen [3], a
dense-array EEG net is placed on the head surface, and small (110 µA) sinusoidal currents are injected into the head volume through selected pairs of electrodes. By measuring
the scalp potential at each of the remaining electrodes, regional head tissue conductivities can be determined
by inverse methods. The obvious concern with this approach is that, since the low skull conductivity causes most
of the injected current to be shunted through the scalp, the sensitivity of the scalp potentials to the inner tissue
conductivities (brain and CSF) is necessarily limited. Despite this physical limitation, we present here an inverse
procedure that can retrieve all four regional head tissue conductivities to within a few percent error.
2. Methods
2.1 Data acquisition
The proposed method of conductivity estimation is designed to be closely integrated with dense-array
EEG data acquisition. Electric current can easily be delivered through any pair of electrodes in the GSN. The Net
Amps system is already capable of injecting small currents into the scalp, and such a method is already being used
at the beginning of data acquisition to estimate scalp-electrode impedance. By injecting sinusoidal current and
averaging over many cycles, the background EEG can be reduced to negligible error. We discuss other noise sources
below.
2.2 Forward problem
To address the main problem posed by the low skull conductivity, we focused here on the
development of effective inverse methods within a spherical head model. Assuming that the frequency of the
injected current is low (f d 1 kHz), the potentials at each time point can be computed as if the current were constant
in time. Standard methods show that the scalp potential is given by
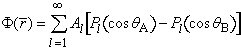
where θA
(θ B) is the angle
between the measurement electrode and the positive (negative) injection electrode [4,13]. The constants Al
depend nonlinearly on the four head radii and the four conductivities σ, and linearly on the magnitude of the injected current I. Taking I = 1
µA results in potentials ranging between ± 50
µV over the scalp surface, depending on
the angle between the injection electrodes. On a 300 MHz Macintosh G3, our numerical implementation of the forward
solution required approximately 1.8 seconds to compute the potentials for a 128-channel net. This code was written
with more emphasis on precision than on speed, however, and we expect that considerable speed increases can be
attained with reasonable effort.
2.3 Inverse problem
Due to the algebraic complexity of the coefficients Al , it is not possible
to solve explicitly for the conductivities σs
in terms of the scalp potentials. Instead, inverse methods must be applied. To guide the inverse solution, we defined
an error function
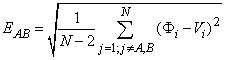
where N is the number of scalp electrodes, Φi and Vi
are the computed and measured scalp potentials, and A and B refer to the positive and negative injection electrodes,
respectively. For a given data set Vi, the best-fit conductivities are those for which EAB
is globally minimum. While it is not possible to visualize EAB in the full
four-dimensional parameter space, it is still useful to visualize it as a function of each of the four conductivities
individually. The different lines in Figure 2 represent electrode separation angles of 32 (dot-dashed), 90 (dashed)
and 180 (solid) degrees.
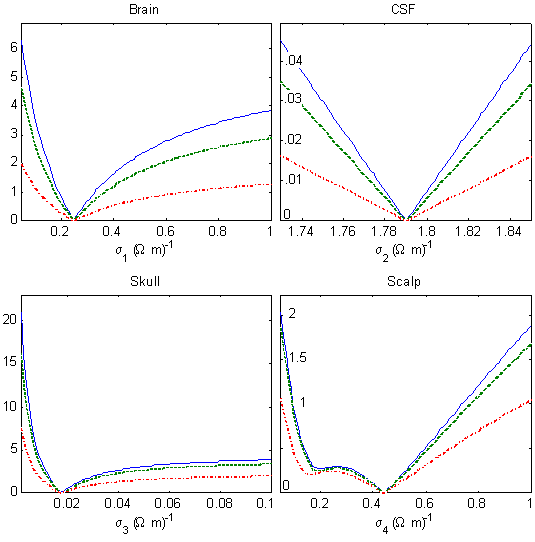
Figure 2. Root-mean-squared error in scalp potentials (µV)
as a function of each head tissue conductivity in a four-sphere model of the human head, assuming 1 µA
injected current.
In each dimension, the error function shows a distinct global minimum. Assuming that this simple structure persists
in the four-dimensional space, the goal of the inverse procedure is to find this global minimum, even in the presence
of inevitable measurement noise. In preliminary studies, we found that retrieval accuracy
was improved by using multiple injection pairs and averaging the corresponding error functions.
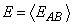
where the average is computed over a specified set of injection pairs. This improvement
is presumably due to the fact that different electrode separation angles generate different current densities in
the head volume, and thereby probe the tissues differently. An additional improvement undoubtedly results simply
from averaging over different instances of measurement noise, although this effect alone is not sufficient to explain
the improvement. In the results described below, we averaged over four injection pairs with electrode separations
equal to 48, 94, 130, and 171 degrees.
2.4 Noise
To minimize the error function E as a function of
the four conductivities σ, we used the
downhill simplex algorithm of Nelder and Mead [9]. For each run of the algorithm, the simplex was initialized randomly
by picking four points normally distributed about their mean values, according to the parameters listed in Table
1. To demonstrate conductivity retrieval in computer simulation, we first generated mock scalp data using the mean
conductivities listed in Table 1. To make the demonstration more realistic, we added noise to the mock data
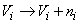
where ni is a zero-mean Gaussian random variable with standard deviation
δV, assumed to be uncorrelated across
electrodes. By averaging over many cycles of the injected current, the background EEG contributes negligible error.
The Net Amps, however, can be expected to contribute noise on the order of δV = 0.1 µV. Errors due to the
misrepresentation of head geometry and electrode placement will be spatially correlated, and are deferred for future
study. In studying retrieval accuracy, therefore, we considered noise levels up to δV = 0.5 µV. Note that increasing
the injected current improves the signal-to-noise ratio in the first two cases only.
3. Results
3.1 Retrieval accuracy
Figure 3 shows the distribution of retrieved conductivities for 50 runs of the simplex
algorithm, assuming the noise level δV = 0.1 µV. Due to the random starting simplex and random noise,
each run of the simplex algorithm yields a slightly different result.
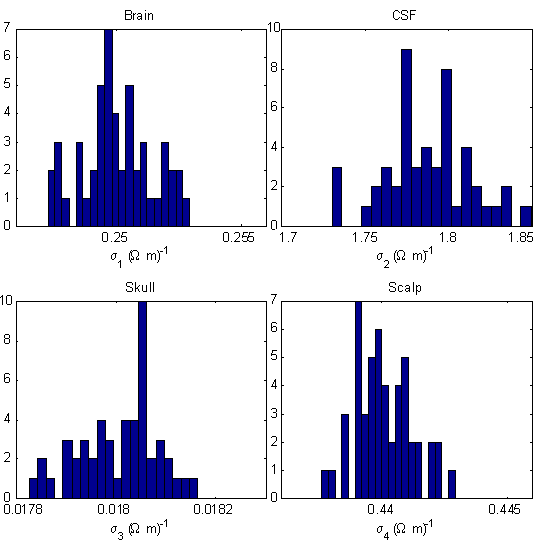
Figure 3. Distribution of retrieved head tissue conductivities in a four-sphere head model,
assuming the injected current I = 1 µA and the noise level δV = 0.1 µV.
Figure 3 shows that, for each tissue, the distribution of results is sharply peaked about
the correct result, and the widths of the distributions are on the order of less than one percent. More precisely,
the corresponding retrieval errors for the four tissues are 0.01 ± 0.6% (brain), 0.13 ± 1.4% (CSF),
0.03 ± 0.43% (skull), and 0.02 ± 0.26% (scalp). This suggests that, despite the low skull
conductivity, it is entirely feasible to measure the average regional conductivity of the brain, CSF, skull and
scalp using scalp current injection.
3.2 Multi-start interpretation
The distributions in Figure 3 can be interpreted in two ways. If the simplex algorithm
were run only once, then these distributions would show the range of retrieval errors. On the other hand, if the
results of the 50 runs are taken together, then the means of the distributions yield much more accurate estimates.
Using the result of many runs in this way can be viewed as a multi-start method, not unlike that used in dipole
source localization [6]. For example, if the means in Figure 3 are taken to provide the true estimate, then this
method produces estimates of regional conductivities with errors on the order of 0.1% for the CSF, and on the order
of 0.01% for the remaining tissues.
3.3 Dependence on noise level
As the noise level increases, the retrieval accuracy degrades, as expected. Even when
the noise level is δV = 0.5 µV,
however, this multi-start method produces a distribution of results for which the error of the mean is still on
the order of only 0.5% for each tissue. Such graceful degradation with noise is a highly sought-after feature of
computer algorithms intended for real-world use.
4. Discussion
We have developed a method of in vivo head tissue conductivity estimation using scalp current
injection. Together with anatomical data from structural MRI, these results can be used to build more accurate
head models, which could substantially improve dense-array EEG data analysis. The fact that the method uses the
same EEG acquisition system makes it both convenient and cost effective. In practice, current injections could
be made when the GSN is placed on the subjects head, and the inverse solution for regional head tissue conductivities
could be computed later during off-line data analysis.
In retrospect, it is easy to understand why this method works. First, since the current source
is known, this inverse problem does not suffer from the issues of non-uniqueness which plague dipole source localization.
Hence the error function appears to have a distinct global minimum in the absence of noise. Second, the addition
of random noise to the data introduces local minima into the error function, making perfect retrieval difficult
or impossible. Practically speaking, however, retrieval accuracy depends upon how severely these local minima distort
the shape, and especially the location of the center, of the global basin of attraction.
For the parameter ranges and noise levels relevant to this problem, the general shape of the error function is
apparently preserved. This allows a clustering of solutions in the vicinity of the correct answer and, by computing
their mean, a very accurate estimate is obtained. As a bonus, the distribution of results generated in this multi-start
approach provides confidence intervals, which would not be available from a single search attempt.
Current research efforts are aimed at using global search algorithms to find
the optimal solution in less iterations, developing boundary element and finite element methods to incorporate
realistic head geometry, and generalizing this approach to detect local changes in tissue impedance which might
signify pathological states of brain tissue.
Acknowledgements
The authors gratefully acknowledge that this work was supported by NIMH 2-R43-MH-53768-02.
References
[1] Awada, K. A., D. R. Jackson, S. B. Baumann, J. T. Williams, D. R. Winton, P. W. Fink, and B.
R. Prasky (1998). Effect of conductivity uncertainties and modeling errors on EEG source localization using a 2-D
model. IEEE Transactions on Biomedical Engineering 45(9):1135-1145.
[2] Baumann, S. B., D. R. Wonzy, S. K. Kelly and F. M. Meno (1997). The electrical
conductivity of human cerebrospinal fluid at body temperature. IEEE Transactions on Biomedical Engineering 44(3):
220-223.
[3] Eriksen, K. J. (1990). In vivo head regional conductivity estimation using a three-sphere
model. Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society
12(4): 1494-1495.
[4] Ferree, T. C., K. J. Eriksen and D. M. Tucker (1999). In vivo head tissue conductivity estimation using scalp current injection.
Submitted to IEEE Transactions on Biomedical Engineering.
[5] Geddes, L. A. and L. E. Baker (1967). The specific resistance of biological materials:
A compendium of data for the biomedical engineer and physiologist. Med. Biol. Eng. 5: 271-293.
[6] Huang, M., C. J. Aine, S. Supek, E. Best, D. Ranken, and E. R. Flynn (1998). Multi-start
downhill simplex method for spatio-temporal source localization in magnetoencephaolgraphy. Electroencephalography
and Clinical Neurophysiology 108: 32-44.
[7] Law, S. K. (1993). Thickness and resistivity variations over the upper surface
of the human skull. Brain Topography 6(2): 99-109.
[8] Luu, P., D. M. Tucker, T. C. Ferree, R. Englander, A. Lockfeld, and H. Lutsep (1999).
Clinical interpretation of stroke-related EEG is enhanced by dense-array spatial sampling: characterization of
stroke-related EEG topography. In preparation.
[9] Press, W. H., S. A. Teukolsky, W. T. Vetterling and B. P. Flannery (1992). Numerical
recipes in C. Cambridge University Press.
[10] Oostendorp, T. and A. van Oosterom (1991). The potential distribution generated
by surface electrodes in inhomogeneous volume conductors of arbitrary shape. IEEE Transactions on Biomedical Engineering
38(5): 409-417.
[11] Laarne, P., P. Kauppinen, J. Hyttinen, J. Malmivuo, and H. Eskola. Effects of tissue
resistivities on EEG sensitivity distribution. Submitted to Medical and Biological Engineering and Computing.
[12] Pilkington, T. C., and R. Plonsey (1982). Engineering Contributions to Biophysical
Electrocardiography. IEEE, New York.
[13] Rush, S. and D. A. Driscoll (1969). EEG electrode sensitivity - an application
of reciprocity. IEEE Transactions on Biomedical Engineering 16(1): 15-22.
[14] Srinivasan, R., D. M. Tucker and M. Murias (1998). Estimating the spatial nyquist
of the human EEG. Behavior Research Methods, Instruments,
and Computers 30: 8-19.
[15] Stok, C. J. (1987). The influence of model parameters on EEG/MEG single dipole
source estimation. IEEE Transactions on Biomedical Engineering 34(4): 289-296.
[16] Tucker, D. M. (1991). Spatial sampling of head electric fields: The geodesic sensor
net. Electroencephalography and Clinical Neurophysiology 79: 413

 Home
Current Issue
Table of Contents
Home
Current Issue
Table of Contents